
Short Summary
The immune system is one of the most intricate and fascinating parts of the human body. In Immune, Philipp Dettmer breaks it down with vivid clarity, guiding readers through the key cells, processes, and microscopic battles that protect us from disease.
Key Takeaways
1️⃣ The Immune System: Self vs. Non-Self
At its core, the immune system is a tool to distinguish self from non-self. Viruses, harmful bacteria, and parasites — all of which can be considered “pathogens” — have been a threat to multicellular life for more than 500 million years, when the first multicellular organisms first appeared. Multicellular beings — like humans and animals — make ideal hosts for these microscopic organisms, who want to invade and take advantage of the resources and nutrients available to them. In order to survive, multicellular beings had to develop sophisticated immune systems to detect and eliminate these threats, which are happening constantly.
The immune system we rely on today is the product of millions of years of evolutionary refinement. Its primary function is to recognize what belongs in the body and what doesn’t. When it identifies something in the body that’s not you — like a virus or harmful bacteria — it attacks it using an army of soldiers and weapons. How does it determine self vs. non-self? If bacteria have been around for billions of years, why haven’t they evolved mechanisms to completely baffle your immune system’s detection capabilities? The answer to these questions lies in the way that bacteria, viruses, and other pathogens are built. Although these microorganisms can build all kinds of different proteins to make themselves look and behave differently to avoid detection, they still need the very basic proteins in order to function. Think of a car — you can mix parts and colors to produce different cars, but you still need wheels and an engine. Your immune cells have receptors that can identify these very basic parts (called Antigens) that are common in most harmful pathogens. A bacteria’s flagella is a good example: the flagella is a tail-like structure made up of proteins that help the bacteria move around. Other than sperm, human cells don’t have a flagella. Therefore, when your immune cells recognize the proteins that make a flagella, they know to kill the organism. This is just one example of how the immune system identifies what doesn’t belong.
Ultimately, the immune system’s goal is to maintain homeostasis — internal balance — within the body. The symptoms you experience during a cold, such as fever or inflammation, are not caused by the virus or bad bacteria itself but by your immune system’s attacks on these bad actors that have invaded your body.
An interesting note about the immune system and pregnancy: when sperm enter a woman’s body, they are recognized as “other” and are attacked by immune system cells like macrophages and neutrophils. Of the 200 million sperm that enter, only a few hundred make it into the fallopian tubes and have a chance to fertilize the egg. Most are eliminated along the way due to the woman’s acidic vaginal environment, immune system, or simply getting off course. This helps explain why getting pregnant is hard.
A similar situation unfolds in the case of organ transplants. The process of installing an organ from one person’s body into another is often very challenging because of the immune system. The immune system labels the new organ, and the cells that make up the organ, as “other” and attacks relentlessly. This is why, unfortunately, many people who receive an organ donation have to spend the rest of their lives taking medication that suppresses the immune system, which leaves them vulnerable to other infections (i.e. bacteria, viruses, cancer).
2️⃣ Cells: Protein Robots
To understand the immune system, you first have to understand cells. We have about 40 trillion cells and 40 trillion helpful bacteria in, and on, our body. About 36 trillion of those bacteria live in our gut and help us digest food; the other four trillion are on our skin, in our lungs and mouth, and in our eye fluid. Cells come in all kinds of types and are jam packed with millions of proteins, which are created when amino acids are linked together in specific sequences. The instructions for assembling these proteins are stored in the DNA found in each cell’s nucleus. Your DNA is a super long strand that consists of smaller sections, your genes, and each gene is the instruction for one protein. The process of building a protein happens in two main steps:
- The DNA is transcribed into a messenger molecule called mRNA
- The mRNA then exits the nucleus and travels to a part of the cell called the ribosome, where it’s translated into a chain of amino acids that folds into a functional protein.
Proteins come in all shapes and sizes, and cells can use them to both build almost anything and communicate with other cells. Think of cells as “protein robots” that are guided by sequences of interactions between proteins. These “protein robots” (i.e. cells) are blind, deaf, and stupid, so how do they know where to go and what to do? Well, cells are covered with receptors that proteins and other things outside of the cell can bind to in order to cause the cell to behave in certain ways. Half of a receptor is inside the cell, half of it is outside the cell. When a certain protein binds to the outer half of the receptor, it sends a signal inside and causes a certain behavior from the cell. For example, when a cytokine — a protein released by immune cells to call for backup at an infection site — binds to the receptor of another immune cell, it steers that cell to the scene. This is the way cells communicate and are able to do things that make sense.
3️⃣ The Two Parts of Your Immune System
Our immune system is made up of two different realms — the Innate Immune System and the Adaptive Immune System.
The Innate Immune System contains all of the defenses you were born with and can be deployed immediately after an invasion occurs. Its weapons are not tailored to any specific bacteria, virus, or any other pathogen — instead, they try to be effective across a wide range of common invaders. For example, it doesn’t have specific weapons against specific types of bacteria like E.coli; but it does have weapons against bacteria in general. The Innate Immune System is your first line of defense and does most of the actual fighting — in fact, most of the billions of soldiers in your immune system are part of the Inmate Immune System. In addition to fighting bad actors, it has to make crucial decisions after an invasion has occurred: How dangerous is this threat? What kind of enemy is attacking? Are more heavy weapons needed? If your Innate Immune System thinks the threat is big enough, it has the power to activate a second line of defense to join the fight: the soldiers of the Adaptive Immune System.
The Adaptive Immune System contains highly specialized super cells that coordinate and support your first line of defenders in the Innate Immune System. Think of these cells as super soldiers that can come to the rescue if the Innate Immune System is having trouble with a particular bacteria or virus and calls for help. Your Adaptive Immune System knows every possible intruder in the universe and has an answer for every single possible microorganism that exists. It possesses the largest library in the universe, and the library is filled with detailed profiles of every current and future possible enemy. Unlike the Innate Immune System, your Adaptive Immune System is not born with these capabilities; they need to be trained and refined over many years, hence the name “adaptive.” In fact, a weak Adaptive Immune System is part of the reason babies and old people are more likely to die from diseases than people in their middle years.
These two realms are interconnected and play off of each other in a beautifully complex way. The Adaptive Immune System is very powerful in its own right, but its main function is to support the Innate Immune System, which is your first line of defense. If a particular bacterium or virus causes too much trouble for your Innate Immune System at the initial site of the infection, it activates The Adaptive Immune System via Dendritic Cells.
4️⃣ Bacteria, Viruses, and Antigens
Most harmful bacteria we encounter aren’t much of a problem. Our immune system recognizes their basic protein parts (Antigens) easily and deals with them without much of an issue every single day. Your real enemies are a small group of elite bacteria and viruses that have found ways to overcome your defenses. These are known as pathogens, a term for any microorganism that can make you sick or cause disease. If something makes you sick — whether it’s a virus, bacterium, fungus, or parasite — it’s considered a pathogen.
Let’s start with bacteria. Bacteria are among the oldest living things on this planet. Emphasis on living; these things are alive, unlike viruses. Like your cells, these are single-celled protein robots that come in a wide variety of shapes and sizes based on their DNA. They have evolved over billions of years and have been very successful on Earth. Bacteria are everywhere! We have 36 trillion in our gut (mostly helpful) and another four trillion on our skin. In one gram of plaque on your teeth, there are more than 7 billion bacteria.
Bacteria view us as a great place to sustain life and reproduce. Because they are single-cell organisms, they can reproduce extremely quickly, which is what makes them difficult for your immune system to deal with. They can also change their genetics at any time, which can also cause problems for our immune system. We are absolutely covered in bacteria — but, fortunately, most bacteria are harmless to us. In fact, some even work with us to keep harmful bacteria away, and trillions of them live in our gut, where they primarily help us break down and digest food. These are “friendly” bacteria. And make no mistake, we wouldn’t be alive without some of these friendly bacteria.
If successful, harmful bacteria that invade us can cause a wide variety of diseases and death. They are especially good at entering your body through cuts, where our protective barrier (the skin) has been breached. Antibiotics have certainly helped us fight bacterial diseases, which used to be one of the leading killers of humans. In 1941, 82% of people who incurred bacterial infections of the blood died. Unfathomably, this means a scratch and a tiny bit of dirt might mean the end of your life. Today, in developed countries, less than 1% of these kinds of infections cause death.
Now let’s discuss Viruses. Unlike bacteria, viruses are dead — there is nothing alive about them. In a nutshell, they are made of a couple lines of genetic code and a few proteins. This is why they must infect living things to stick around. Infecting living things is their No. 1 goal, and if all living things on the planet died, they would die as well because there would be nothing to infect. They can’t reproduce or do anything without first infecting a living thing, like a human cell. Unbelievably, there are actually billions and billions and billions of viruses in the world, but only about 200 of them have the proteins necessary to attach to human cells and infect us. Thank goodness.
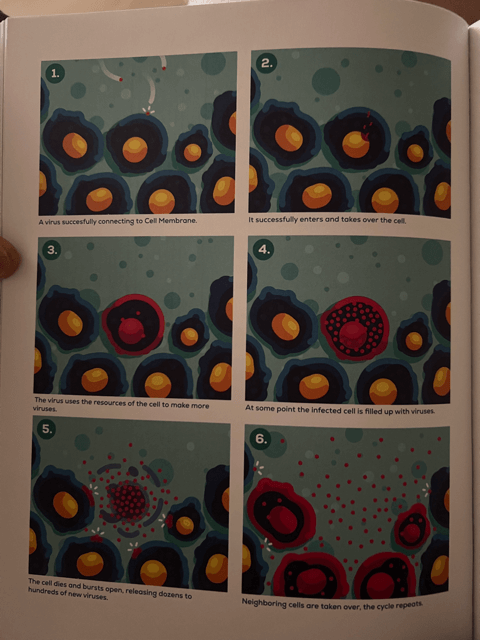
How does a virus infect us? First it gets inside of us. Rather than breaking through the skin, the most common way it gets inside of us is through our lungs as we breathe in air that a virus is hanging out in (think about someone with the flu coughing near you). Once inside, it usually binds to one of the Epithelial Cells that line the protective mucous layer inside our lungs and takes the cell over by transferring its genetic material. This transfer of genetic material turns the cell into a virus-producing machine. At some point, the cell is completely filled up with viruses. The cell then dies and bursts open, releasing hundreds of new viruses that go out and repeat this same process with cells nearby. Before you know it, the virus has become a raging wildfire inside of your body. And this is a defining feature of these things: Nothing multiplies, and mutates (changes), as fast as a virus does.
Influenza (the flu) and COVID-19 are examples of viruses. The Spanish Flu killed 40 million people in 1918-19. Because viruses like these mutate and multiply so quickly — and because their tactics are different than bacteria — the immune response to a bacterial infection and a viral infection are much different.
Whether it’s harmful bacteria, a virus, a parasite, or even cancer, these pathogens all have one thing in common: they want to destroy your cells. If they manage to get inside, multiply, and spread, they can hijack or kill enough cells to overtake your body, which manifests as serious illness or even death. The immune system’s mission is to protect your cells — and therefore, protect you — by detecting, attacking, and eliminating anything that poses a threat. In this way, every illness or disease comes down to what’s happening with your cells.
How does your immune system recognize and begin attacking these pathogens? Every pathogen — whether it’s bacteria or a virus — has something called Antigens, which are basically small pieces of protein on the surface of the pathogen that serve as “tiny ID tags.” Antigens are really, really important for two main reasons: (i.) they are what your immune system uses to identify exactly what the pathogen is, and (ii.) your Adaptive Immune System uses the Antigens to activate and make the correct Antibodies, Helper T Cells, and Killer T Cells that can lock onto and destroy that specific pathogen. Even our own cells have these “tiny ID tags,” and when our immune system weapons accidentally lock onto our Self-Antigens, it usually leads to Autoimmune Diseases.
5️⃣ The Desert Kingdom of the Skin & The Swamp Kingdom of the Mucosa
For a harmful bacteria or pathogen to become a problem for you, it first has to find a way to break through your skin. Other than your gut, which is made for and ruled by about 36-40 trillion helpful bacteria that your body has welcomed in, the skin is the second most populated place on your body in terms of bacteria. You have about 10 billion bacteria on your skin at all times, some good and some bad. Fortunately for us, our skin is an outstanding protective barrier. Think of it as a dead, salty desert that’s hostile to most microbes, or The Desert Kingdom of the Skin.
The skin is basically made up of hundreds of layers of skin cells that are constantly being regenerated by our skin stem cells. The skin stem cells lie underneath, or deeper in the epidermis, and crank out new skin cells, which form the very bottom layers of your skin and slowly move up over time. As they are pushed up, these living skin cells get coated in a fatty layer of “passive antibiotics” that protect you from harmful enemies. By the time the skin cells reach the surface layers, they’re dead. All of the skin you see in the mirror is dead skin, made up of up to 50 layers of dead skin cells. As this dead layer of skin is damaged and used during your daily life, it is constantly being shed and replaced by new skin cells that are moving up from below. It takes skin between 30-50 days to completely turn over, and you shed about 40,000 dead skin cells every second! That’s important because when your dead skin flakes away, harmful bacteria crawling on it go with it. When you wipe away some dust, a lot of what you’re wiping up is your own dead skin cells.
The skin also becomes salty when you sweat, which harmful bacteria don’t like. As mentioned earlier, we have 10 billion bacteria crawling on our skin at all times — most of these bacteria are not harmful and actually do us a favor by taking up real estate from harmful bacteria. Finally, another great benefit of the skin is that it’s basically immune to viruses, which can only infect living cells — and the surface of your skin consists only of dead cells. Nothing to infect here!
Overall, the skin is an outstanding barrier. In fact, the skin is so effective as a defense perimeter that pathogens usually don’t have much of a shot at getting inside us here, as long as we don’t cut ourselves and open up an opportunity. Instead, most infections that we deal with throughout our lives enter the body elsewhere, like the nose, mouth, eyes, lungs, digestive tract, and reproductive tract. These are the most vulnerable areas on your body because they are where your insides directly interact with the outside world (i.e. breathing in stuff, eating food, etc.). All kinds of stuff that isn’t YOU comes into your body from these areas. As protection, they are lined with what is called Mucosa — or a mucous layer. Think of this as a giant swampland that provides a layer of protection for our insides. It’s the “skin” of our insides. We can call it The Swamp Kingdom of the Mucosa.
The immune system has to behave differently in these mucous layers than it does when you have a breach of the skin. Why? It can’t afford to launch a full-blown immune response to every little thing that comes into your body from breathing or eating, for example. It has to walk a fine line between being too aggressive and too relaxed. If it attacked every little thing that entered, you would have constant inflammation. Not good.
We think of mucous as only being in the nose, but all of these vulnerable areas — the mouth, nose, eyes, respiratory tract (lungs), digestive tract (gut), and reproductive tract — are lined with a layer of it. Just like the skin, mucous contains built-in defense systems that make it difficult for harmful bacteria to get deeper inside of us. For one, the stuff is slimy and is very difficult for bacteria to swim through. Second, it’s loaded with salt, enzymes, and special substances that starve bacteria to death by sponging up critical nutrients that they need. Third, it is saturated with special Antibodies that are ready to “mark” harmful bacteria for destruction by other immune cells if needed.
Depending on the location, mucous behaves a little differently — the mucous layer in your lungs has a very different job than the mucous layer in your gut, and so on. Because it’s fascinating, let’s look at the gut (intestines) as an example. When you eat something, your saliva starts to break it down. After the food is swallowed, it heads to the ocean of acid in your stomach, which breaks the food down and is also where many bacteria on the food die due to being submerged in literal acid. After the stomach, the journey continues to the intestines, where 90% of the nutrients you need to survive are absorbed. Here in the gut is where we find about 36 trillion bacteria that are essential to our survival — they help us digest the food further and produce vitamins that we can’t produce ourselves.
Because of all that bacteria (some good, some bad) that sit directly above the mucous layer of your gut, the immune system of the gut acts semi-independently of the immune system guarding the rest of your body. The mucous layer of the gut is always being breached by some of those bacteria, and your immune system has to constantly determine friend or foe. It does this by having immune cells like Macrophages, B Cells, Dendritic Cells, and Antibodies that behave a little differently than they do in other areas of the body. I won’t get into the details. But together, these cells and Antibodies kill a lot of bacteria in the gut that try to cross the mucous layer — in fact, around 30% of your poop consists of dead (or still alive) bacteria. Also, the last thing you want is a big immune response that leads to inflammation in the gut. Inflammation means more fluid in the gut, which is what causes diarrhea.
Another interesting area to look at is the lungs, where a mucous layer lines the respiratory tract and protects you from all kinds of things. The lungs are where you are most vulnerable to infection from a virus, although bacteria can get in here as well. That’s because you inhale a couple thousand gallons of air through your lungs every day, and there are all kinds of virus, bacteria, fungi, and other stuff hanging out in the air around you. Think about when someone with a cold coughs near you — the stuff in that cloud of air moves quickly and some of it can get into your lungs. Overall, some of the most dangerous pathogenic viruses use the lungs as their main entry point.
6️⃣ An Immune System Response: Bacterial Infection
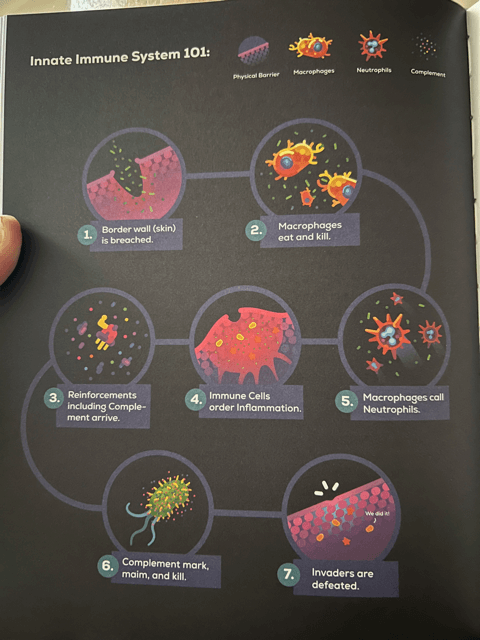
Say you cut your knee sliding during an adult softball game. What happens when your skin is opened up and harmful bacteria on you and in the surrounding environment are able to walk right in? To help understand what happens during a bacterial infection, it’s helpful to look at the body as a huge continent with trillions of civilians (cells). A war breaks out, and the cells of the Innate Immune System are the first soldiers sent to fight the enemy. Below is a very simple look at how your Innate Immune System works while fighting a bacterial infection. The immune system’s process for fighting a virus is different and will be covered in a separate takeaway.
- Cytokines — The damage caused by whatever caused your skin to open up kills many of your civilians right away. When your immune cells come across other dead cells or harmful bacteria, they raise the alarm by releasing Cytokines, which are small proteins that convey information to other cells. The cytokines carry the message: “Danger! Enemies are around! Come help!” Immune cells like macrophages and neutrophils “smell” the cytokines and follow their trace to the battlefield. The more cytokines that are released by your immune cells, the bigger the threat. In rare circumstances, your immune cells will release tons of cytokines when the battle is over or when there’s no threat at all. Because cytokines signal the need for an immune response, your cells go crazy attacking things for no reason, eventually causing inflammation and tissue damage. This process is common in autoimmune diseases, where your immune system mistakes you for the enemy.
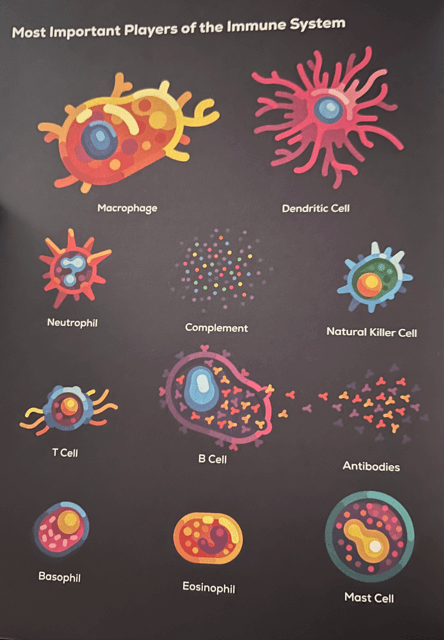
- Macrophages — Your Innate Immune System responds to the Cytokines immediately by sending Macrophages, the largest immune system cells in the body, to the scene. These cells are huge, much bigger than the average cell — if the normal cell is a human, a Macrophage is a rhino. Macrophages, known as Great Eaters, eat dead cells and living bad bacteria, clear debris from the battlefield, and start the repair process. They swallow as many enemies as they can, but they can’t eliminate the threat by themselves. They release more cytokines and call for backup.
- Neutrophils — Neutrophil cells travel from the blood to the scene to assist. The most abundant immune cell in your blood, they immediately begin hunting and eating bacteria, but they’re much less careful about it because they die fairly quickly. They are the “crazy suicidal Spartan warriors” of the immune system. They eat, throw acid, and kill themselves to create deadly traps. Their goal is to go as hard as they can, and they’re not too concerned about damaging helpful allies. Many of them explode when they’re done, and the chemicals they release are harmful to bad bacteria. Our own healthy civilian cells are sort of afraid of Neutrophils because they are so aggressive. Every day, even without an infection, 100 billion of these warrior cells voluntarily die and are immediately replaced with new ones. The puss that oozes from your cut is the dead bodies of millions of Neutrophils that fought to the death for you, mixed in with dead civilian cells and bacteria.
- Platelets — While all of this is going on, the wound on your skin has developed a bit of a crust. This is your platelets closing it off. Platelets are blood cells that lump together and form a sticky substance that clogs the wound. Their goal is to stop blood loss and prevent more intruders from entering. This enables fresh skin cells to slowly start closing the hole.
- Inflammation — Macrophages and Neutrophils fighting at the scene, as well as cells that have been killed in battle, send signals that cause inflammation, which is a crucial defense process and causes the area around your cut or injury to swell up. Inflammation occurs when your blood vessels open up and let warm fluid stream into the battlefield. Imagine this as floodgates opening up, where blood carrying all sorts of helpful immune cell reinforcements comes rushing in very quickly to defend the injured or infected area. When this happens, the extra fluid causes you to experience some swelling in the area. Inflammation is the universal response of your immune system to any sort of breach or damage. No matter if you burn yourself, cut yourself, or get a bruise. No matter if bacteria or viruses infect your nose, lungs, or gut. Damage or danger — perceived or real — causes inflammation. Inflammation is the red swelling and itching from an insect bite, the sore throat when you have a cold. Interestingly, chronic inflammation is terrible for you; in fact, it’s an underlying cause of about 50% of deaths today. Inflammation is also a big reason for many COVID and pneumonia deaths; when the virus infects people’s lungs, it causes a rush of fluid to the area, and the swelling makes it hard to breathe and get enough oxygen. But in the context of a typical immune response, inflammation is very helpful in three main ways:
- It restricts an infection to an area and stops it from spreading
- It helps remove damaged and dead tissue
- It serves as an expressway to get your immune cells and attack proteins directly to the site of infection
- Complement Proteins — This rush of fluid to the scene caused by inflammation carries a silent killer into the battle zone: Complement Proteins from The Complement System. These proteins are super tiny and float passively in your blood until an infection occurs and one of them is activated. When one of them is activated at the scene of an infection, it causes a chain reaction that leads to the activation of others around it. Soon, an army of millions of these proteins are activated and become a weapon for your immune system. What do they do? Three things: (i.) They “mark” bacteria by coating their surface, making it easier for your Macrophages and Neutrophils to locate, grab, and eat them; (ii.) they call for backup by sending chemical signals to recruit more immune cells to the area; (iii.) they literally “rip holes” in the bacteria, causing them to “bleed out.” Complement Proteins play a big role in creating a very fast Innate Immune System response. Where there’s inflammation, there’s Complement Proteins.
In most cases, this is where the battle ends. All bacteria are killed. Your cut and infection heal up. All is well. But we’ll assume a certain species of harmful bacteria is somehow finding a way to reproduce and evade your Innate Immune System soldiers. Your Macrophages and Neutrophils keep fighting, but the inflammation continues to rise and huge numbers of civilian cells are dying. What happens now? This is when the Adaptive Immune System gets involved. Below is a very high-level look at how this system responds (more detailed information about how some of this unfolds is in the next takeaway):
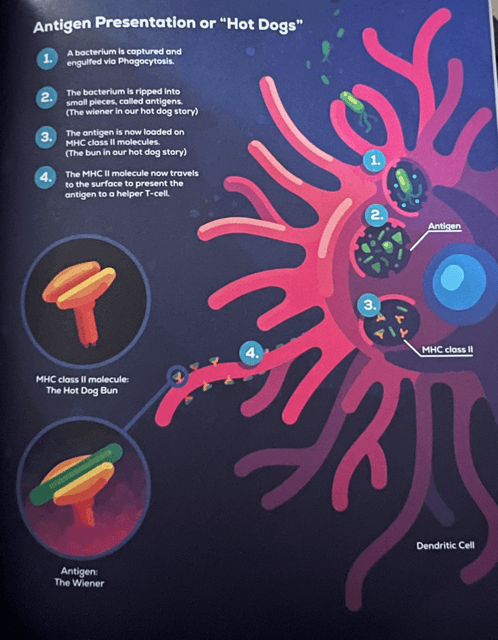
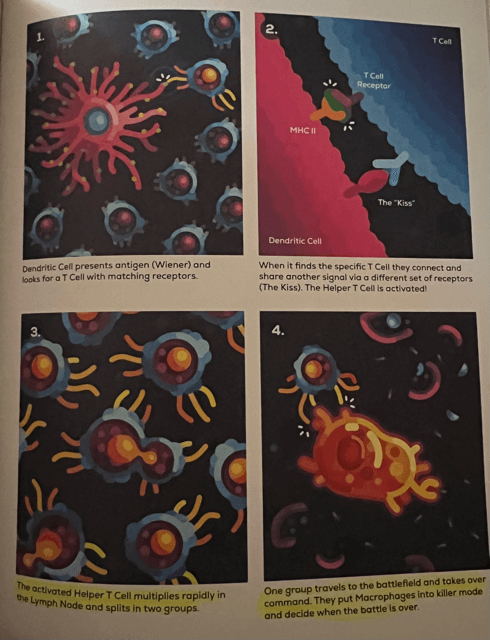
- Dendritic Cells — When a certain species of harmful bacteria is able to evade your immune system and reproduce quickly in your body, Dendritic Cells have to make a decision about whether or not to call for help and activate the very powerful Adaptive Immune System. Dendritic Cells belong to the Innate Immune System, and they are crucial. As the initial battle at the scene of an infection is taking place, they lurk in the background and assess the situation. They go around and take “samples” of dead enemies. This process involves crunching a bacterium into a pile of Antigens, which are basically the proteins it’s made up of.
- MHC Class II Molecules — Once the bacterium is reduced to a pile of antigens, Dendritic Cells load the antigens onto a molecule that lives inside them called MHC Class II. These molecules look like an empty hot dog bun, and the antigen is loaded onto them like a wiener. Once loaded with wieners (antigens), the MHC Class II molecules move to the surface of the Dendritic Cell so they can “present” the bacteria’s antigens to Helper T Cells of the Adaptive Immune System.
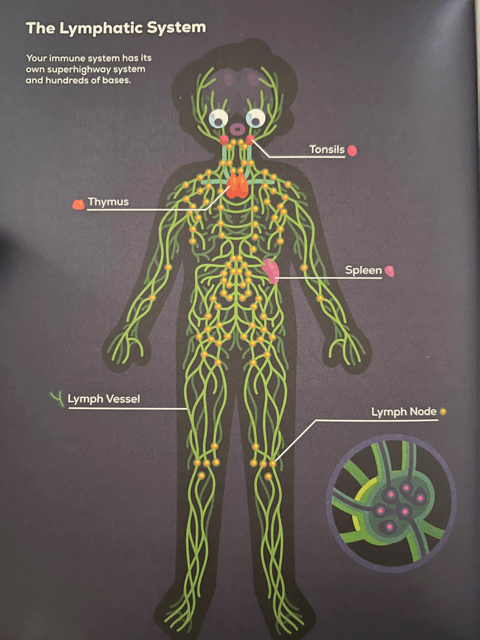
- The Lymphatic System — But first, the Dendritic Cells have to get to the Helper T Cells using the Lymphatic System. For cells, the human body is an enormous continent. The Lymphatic System provides a way for your immune cells to traverse this huge continent. This system is primarily responsible for draining excess fluid called Lymph from your muscles and tissues and delivering it back to the blood so it can recirculate. This process prevents you from swelling up like a balloon over time as the battle is waging. It transports lymph using a network of superhighways (vessels) that your immune cells can also use to travel through the body, and it’s the system the Dendritic Cells use in this case to transport their antigen samples to the Helper T Cells.
- Lymph Nodes — Located throughout your body are about 600 “megacities” called Lymph Nodes. Lymph must pass through these megacities before it can rejoin blood circulation. The lymph nodes are also where immune cells from the Innate Immune System, like Dendritic Cells, meet up with immune cells from the Adaptive Immune System, like Helper T Cells. In this scenario, the Dendritic Cells take their MHC Class II antigen hot dogs to the nearest lymph node stations to find Helper T Cells that have receptors that match the bacterium’s antigens. The exact receptor for the particular antigen that the Dendritic Cells are presenting is not always found right away. Instead, Helper T Cells will literally rearrange their genes to manufacture a receptor that matches the exact antigen of the harmful bacteria.
- T Cells and Antibodies — Once Helper T Cells with the right receptor for the specific antigen wieners it is carrying, more of them are created. Additional weapons built specially to hunt and kill the particular bacterium — like Antibodies and Killer T Cells — are also created. Once created in the lymph nodes, Helper T Cells race over to the battlefield and use special cytokine signals to motivate extremely tired Macrophages. It’s as if they give these soldiers a second wind; a jolt of energy that inspires them to get back to ferociously killing bacteria. Not long after, millions of Antibodies and Killer T Cells arrive on the scene. Antibodies are super soldiers created by B Cells that act as assassins specifically designed to eliminate the exact species of bacteria or virus in your system. They do this primarily by “marking” the enemy for destruction by other immune cells. What was once a brutal battle for days now turns into a one-sided slaughter, as your Adaptive Immune System comes to the rescue.
- Healing — If all goes well, this is where the battle ends. The last bacterium is eaten, Neutrophils and Helper T Cells begin to kill themselves now that victory has been won, Macrophages clean up the debris by eating dead cells before also killing themselves, excess fluid is carried away through the Lymphatic System, inflammation subsides, and young civilian cells take the place of the fallen. Healing is happening. From a human perspective, you notice that the swelling is gone and the wound, injury, or cold feels better.
- Immune: Memory Cells — A critical final piece to this sequence is Memory Cells, which are created after the war has been won. When you hear that you’re immune to a disease, it means you have living Memory T Cells and Memory B Cells roaming around your body that “remember” a specific enemy’s antigens and can easily eliminate the threat if it ever returns later in your life. This is why you normally will not suffer the same sickness or disease twice.
7️⃣ The Adaptive Immune System
The Adaptive Immune System is extremely powerful and complex, and it deserves a closer look as its own takeaway. As mentioned earlier, the Adaptive Immune System contains specialized super soldiers that can help the Innate Immune System deal with a particular species of harmful bacteria or virus that is reproducing quickly and causing problems. The warriors of this system do not come to the battlefield of an infection right away; the Innate Immune System soldiers head to the scene first while Dendritic Cells assess the situation. If the Innate Immune System struggles enough, Dendritic Cells will call on the very powerful Adaptive Immune System for help by taking the pathogen’s antigens to Helper T Cells and B Cells located in your lymph nodes.
Why is our Adaptive Immune System so important? Bacteria have been around for billions of years and possess two features that pose big problems for our immune system: (i.) they are single-cell organisms, which allows them to reproduce extremely fast, and (ii.) they can literally change their own genetics/proteins at any time to evade Innate Immune System soldiers (e.g. Macrophages and Neutrophils). Most bacteria on and around you are harmless or even helpful to you. But these unique abilities of bacteria become a big issue when harmful bacteria looking to take you out get into your body (e.g. E.coli). Your Innate Immune System soldiers (e.g. Macrophages and Neutrophils) usually don’t have any problem recognizing the antigens (i.e. proteins) on these harmful bacteria and killing them; they do it every day without batting an eye. But when a particular species of harmful bacteria possessing antigens that the Innate Immune System is not familiar with gets in and begins reproducing and changing genes, it needs help before things get really bad for you in the form of disease. These bacteria evade detection because they possess antigens (proteins) that the Innate Immune System is not familiar with.
Viruses are arguably even worse. They are able to hide inside of your cells, and they multiply explosively. The Innate Immune System is not very effective at fighting viruses — the best it can really do is buy some time for more effective weapons.
That’s where the Adaptive Immune System comes in. This system has an incredible superpower: it can generate millions of different immune cells, each with receptors specifically designed to recognize specific antigens — which are protein pieces from a virus, bacterium, or other invader. Together, these cells form the largest biological “library” in your body and give you the ability to make weapons for any pathogen the world might throw at you. Even for new threats like COVID-19, your Adaptive Immune System can either find or create (via mixing and matching genes) immune cells with the right receptor — it just takes time. That’s why it’s called “adaptive”: it learns and responds to each new invader, building immunity that can protect you in the future.
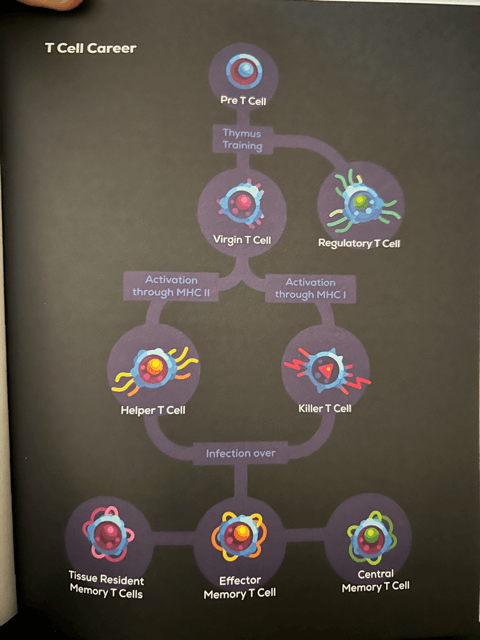
Arguably the most important infantry of soldiers in this system are T Cells, which are born in the Bone Marrow but are trained for duty in the Thymus (hence the name “T Cells”). Resting a few inches under your throat, the Thymus is one of the most under-appreciated organs in the human body. One of its big responsibilities is training T Cells; it’s a giant T Cell training academy. T Cells — Both Helper T Cells and Killer T Cells — are born with receptors capable of binding to the antigens (proteins) of bacteria and can mix and match their genetic code to connect to every possible antigen in the universe. This is why you have the ability to recognize and fight any infection known to man.
During an infection, Dendritic Cells from the Innate Immune System can activate the Adaptive Immune System by finding a Helper T Cell that has the exact receptor for the harmful bacteria’s antigen that it is carrying (the MHC Class II “hot dog” from the previous takeaway). When this happens, the Helper T Cell clones itself in your lymph nodes until there are thousands of them ready to fight the bacteria that is causing problems (this is why your lymph nodes swell during a cold). Then they split into two groups. One group heads to the battlefield and takes command. They put tired Macrophages into killer mode using chemical signals. They coordinate things between other fighters. This happens about 5-7 days after the infection. The second group remains in the lymph nodes to help activate your B Cells, which are your living weapons factory.
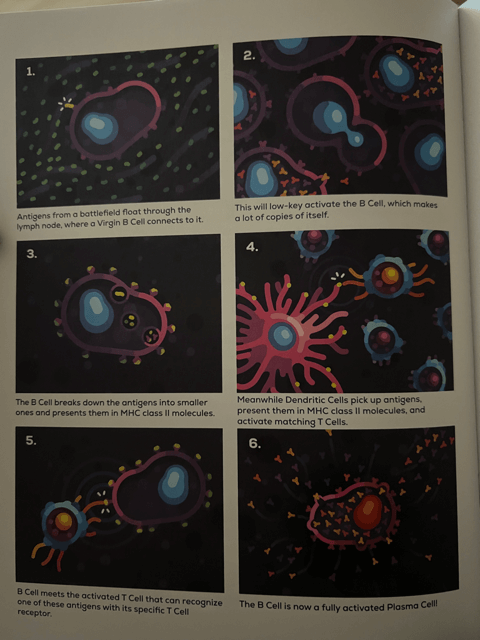
B Cells originate in the Bone Marrow and are similar to T Cells in that, combined, they have millions of different receptors for millions of antigens that a harmful bacteria might possess. What makes B Cells special and very dangerous is that they produce the most lethal specialized weapon in the immune system: Antibodies. These are receptors on the surface of B Cells that can also be used as weapons against specific antigens that a pathogen possesses. B Cells must undergo rigorous training in the Bone Marrow and are then released into your Lymphatic System in an inactive state. B Cells can only become activated using a two-factor authentication process:
- Step 1 — B Cells hang out in your Lymph Nodes, the “megacities” of the Lymphatic System “superhighway” network. Lymph — which carries away from the battlefield excess fluid from inflammation, antigens (proteins) from dead enemy bacteria, and other “garbage” — passes through the Lymph Nodes. B Cells basically sift through the Lymph looking for antigens that match their receptors. If they find an antigen that matches one of their receptors, they know that there’s an enemy in the body and begin making clones of the exact B Cell with the specific receptor that matches the enemy’s antigen. These cloned B Cells then begin making limited quantities of basic Antibodies while they await the second step.
- Step 2 — Without a second step, these B Cells from step one would die in about a day, figuring the infection must not be that bad. A B Cell becomes FULLY activated when it finds an activated Helper T Cell presenting the same enemy antigen. When this happens, the B Cell knows for sure that something bad is going on and turns into a Plasma Cell that generates highly specialized Antibodies. The Plasma Cell pumps these out at a rate of about 2,000 per second — millions of them are created and sent to the battlefield through the blood and Lymphatic System to fight the pathogen that is causing problems for you. This is one of the most powerful stages of the Adaptive Immune System and can turn the tide of an infection quickly.
So, what are Antibodies? They are your super weapons, the main reason you can survive serious infections. Antibodies are essentially copies of the B Cell’s receptor, designed to stick to one specific antigen on an enemy and flag it for destruction. Think of antibodies as proteins that act like guided missiles. They can neutralize viruses by preventing them from infecting your living cells and “marking” bacteria for cleanup by other immune cells like Macrophages and Neutrophils. Antibodies are tiny in structure and look like little crabs with two pincers and a “cute little butt.” The pincers “pinch” the pathogen’s antigens with a death-grip, and their “cute little butt” makes it easy for other immune cells like Macrophages and Neutrophils to latch on and kill the pathogen. Some Antibodies can team up and bind to multiple pathogens, creating big clumps of enemy bodies ready for execution. This makes it nice and easy for your other immune cells to score “double kills” and “triple kills.”
Killer T Cells are also activated via a two-step verification process and are critical when fighting a virus. Viruses like to hide and replicate inside of your cells. They then cause the cell to burst open, and the viruses go out and repeat the process with other nearby cells. In short, Killer T Cells are able to inspect each of your cells and look inside of them. If they see virus proteins inside the cell, they order the cell to kill itself gently. The cell then folds up, trapping the virus inside, and Macrophages come by to eat the virus.
Once all harmful bacteria are destroyed and the infection is over, most Helper T Cells kill themselves, while a small group of them become Memory T Cells. When you hear that you’re immune to a disease, it means you have living Memory T Cells and Memory B Cells roaming around your body that “remember” a specific enemy and can easily kill it if it ever returns later in your life. This is why you normally will not suffer the same sickness or disease twice.
8️⃣ Underappreciated Organs: The Thymus and Spleen
One of the real dangers of the immune system is that the soldiers that fight your enemies might turn on you. This is a particular concern for T Cells and B Cells. Fortunately, there are “academies” that these cells must pass before they are cleared for duty.
For your body, the worry with both of these powerful cells is that they might be born with a receptor that binds to, and kills, your own friendly cells. This is the cause of most Autoimmune Diseases — our Adaptive Immune System thinks our own cells are enemies and attacks us. For T Cells, the Thymus helps prevent that. It’s an academy that newly born T Cells must pass before being put to use. The biggest exam at the academy involves testing a T Cell’s ability to recognize and connect to the proteins (Self-Antigens) on the surface of our own cells. If it can, it is killed. All in all, 98 of 100 T Cells are killed; just 2% pass the Thymus academy and are cleared for duty. This amounts to about 20 million T Cells cleared to enter your bloodstream and Lymphatic System to fight infections. The dead cadets are eaten by your Macrophages. The Thymus is always shrinking, and by about age 85, it’s almost entirely inactive. Once it’s gone, your body struggles to produce new T Cells, which is a huge hit to your immune system. This is one of the reasons older adults have a hard time battling infections, disease, and cancer.
B Cells must also pass rigorous training before they are cleared for duty and allowed to patrol the body. Their training happens in the Bone Marrow, which is the same place they are born. The training curriculum is very similar to what T Cells go through — the most important test is that they cannot bind to the proteins and molecules on the surface of your own friendly cells. If they prove during training that they can, they are killed.
The Spleen is another under-appreciated organ. It’s basically a large Lymph Node about the size of a peach that does all of the things that your 600+ Lymph Nodes do — but instead of filtering Lymph, its focus is on filtering blood. The Spleen is where your old blood cells are filtered and recycled when their life comes to an end. Your Spleen also stores an emergency reserve of blood, about a cup, which is critical if something bad happens and your body needs extra blood. And that’s not all — it also serves as one of the centers of your immune system soldier cells, a sort of barracks. The Spleen should get more love than it does!
9️⃣ An Immune Response: Viral Infections
Our immune system can’t rely on the same weapons to fight a viral infection as it uses to fight a bacterial infection. A virus is smaller and usually a lot more difficult to detect than bacteria. They also hide inside of living cells and try to manipulate infected cells to trick the immune system into standing down. Think back to the ancient Greeks and their invasion of Troy. Bacteria are like the warriors who stormed the gates — loud, aggressive, and ready for battle out in the open. Our immune system can usually recognize the threat and handle it fairly easily. Viruses, on the other hand, are like the soldiers hidden inside the Trojan horse — silent, sneaky, and waiting to strike from within once they’re let inside. Viruses can also mutate (change) and multiply much, much more quickly than bacteria.
Pathogenic viruses are truly scary enemies. They hide inside civilian cells and can multiply explosively. At the height of a virus, you can have billions of viruses inside your body. As a result, your immune system has to approach viruses differently than bacteria. It’s complicated, but here’s a simplified and general look at what happens:
- Entry — A virus, like the flu, enters the body usually through the lungs and the air we breathe in. All kinds of viruses and bacteria hang out in the air around us and can enter us this way. The lungs are one of our most vulnerable and exposed areas. As mentioned previously, viruses find it almost impossible to break through the skin.
- Hostile Cell Takeover — Once inside, most viruses die in the mucous layer lining our lungs. But occasionally one (all it takes is one) is able to bind to one of the Epithelial Cells that lines the mucous layer and performs a “hostile takeover” of the cell by transferring its genetic material. This transfer of genetic material turns the cell into a virus-producing machine. Eventually, the cell is completely filled up with viruses. The cell then dies and bursts open, releasing hundreds of new viruses that go out and repeat this same process with cells nearby. If an immune response doesn’t happen quickly, the virus can become a raging wildfire inside of your body.
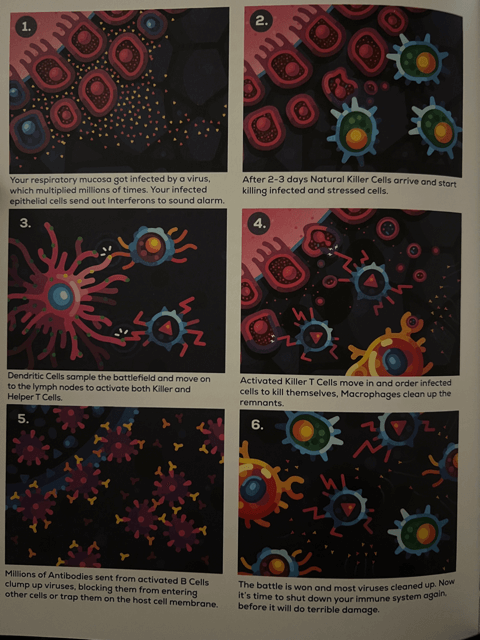
- Cytokines & Interferons — At this point, the Adaptive Immune System is desperately needed — but it takes time for that system to get going. The Innate Immune System has to buy some time and prevent the virus from spreading like a wildfire. Epithelial Cells that realize they are being invaded by a virus do this by releasing Cytokines, which act as signals for backup. They specifically release a type of emergency Cytokine called Interferons, which warn other cells to shut down protein production. This slows down the spread of the virus considerably by making surrounding cells more aware and resistant to infection.
- Plasmacytoid Dendritic Cells — Viruses love to stay hidden from the immune system, but there comes a point where that’s not possible anymore. When cells die an unnatural death, they release all kinds of Cytokines that warn the rest of the immune system that danger is present. So as more and more cells die from the virus, more Cytokines are released and more inflammation is triggered. Plasmacytoid Dendritic Cells aid this warning process. These are a special type of Dendritic Cell that are very good at detecting viruses and pump out huge amounts of Interferons. This helps warn the rest of the immune system and tells nearby cells to shutdown protein production, which slows the virus down.
- Macrophages and Neutrophils — At this point, a full-blown war is on. Macrophages and Neutrophils of the Innate Immune System rush to the scene and begin doing what they can. But the Innate Immune System is not very good at fighting viruses; it can only really slow the virus down, not stop it. The Adaptive Immune System is desperately needed.
- Pyrogens and Fever — As the war wages on, your immune cells begin to release another kind of Cytokine called Pyrogens that act as a more serious call for help. Pyrogens are chemicals that cause Fever. They tell the brain to crank up the temperature. Why is a Fever important while fighting any kind of pathogen? It creates a body-wide heating response that makes life much harder for pathogens while also enabling your immune cells to fight harder. Think about running outside — it’s much harder to run in warm or hot temperatures versus nice, crisp, cool temperatures. The same thing takes place for pathogens in your body when a Fever occurs. Note — a Fever of 104 degrees Fahrenheit is very dangerous and you should seek immediate medical attention. Your brain cranks up the heat and induces a Fever in two main ways:
- Shivering — Your brain induces shivering, which is just your muscles contracting really quickly. These quick contractions generate heat as a byproduct.
- Closing Blood Vessels — The blood vessels close to the surface of your body contract which reduces the heat that can escape through your skin. This is why you usually feel so cold while being sick or having a Fever. Your brain and body are trying to heat up your core to make things unpleasant on the pathogen.
- Additional Physical Symptoms — The war being fought inside you manifests as the typical symptoms of a flu: Fever, muscle aches, runny nose, sore throat, cough, feeling cold, etc. When you blow your nose, the more colorful your snot, the more Neutrophils have given their life. Another symptom is fatigue and lack of appetite. These symptoms are your body trying to tell you to conserve energy so it can fight the pathogen. An immune response takes a ton of energy. So does digesting food and doing anything but laying in bed. To conserve energy for the fight, you should try to obey these signals by resting and not eating too much.
- MHC Class I Molecules — Back to the battle. Viruses spend most of their time hiding and multiplying inside of your living cells. This is why they are so dangerous and hard to stop. To stop the virus, your immune system has to be able recognize, then kill, your own cells that have been infected. How does this happen? The answer is Major Histocompatibility Complex Class I Molecules (MHC Class I). These molecules act as “display windows,” showing what kind of proteins and antigens are being made inside of a cell. As discussed earlier, cells are filled with millions of proteins. When a virus infects one of your cells, it begins creating virus proteins and antigens. It’s complicated, but MHC Class I molecules basically allow your immune cells to go up to any cell in your body with a nucleus (so, all but red blood cells), peer into a little “display window,” and see what kind of proteins are inside. If the proteins inside look foreign, the cell is deemed infected and killed on the spot.
- Killer T Cells — Killer T Cells are among the most powerful cells in your immune system, and they depend on the MHC Class I molecule described above. Dendritic Cells of the Innate Immune System are able to activate Helper T Cells and Killer T Cells. They have this capability because they are able to present a pathogen’s antigens in both MHC Class I and MHC Class II molecules. The MHC Class I molecule (display window) helps the Dendritic Cell activate the Killer T Cell with the right receptor for the pathogen’s antigens, while the MHC Class II (the hot dog) helps it locate and activate the Helper T Cell with the right receptor.
- “Serial Killing” — Once activated, the Killer T Cell clones itself then heads to the infected area to evaluate your cells. It goes up to the MHC Class I “display windows” on every one of the cells in the area and peers inside. If what it sees inside is not right, it deems the cell infected and tells the cell to kill itself in a controlled manner. By dying in a controlled manner, the virus is carefully trapped inside the dead cell, and Macrophages come by to eat everything up. This T Cell murder spree happens at a mass scale — usually about 10 days after the battle started — as it quickly takes out your infected cells in a process known as “serial killing.”
- Natural Killer Cells — Viruses aren’t stupid. They know that Killer T Cells use MHC Class I molecules to peer inside infected cells and decide whether or not to kill them. As a result, viruses found a way to prevent this “display window” from appearing outside the infected cells, allowing them to stay hidden from Killer T Cells and other immune cells. Enter Natural Killer Cells. These are siblings of Killer T Cells that arrive at the battleground within 2-3 days of infection. Their job? To hunt cells infected by viruses and cancer. How do they do this? They check to see if a cell has MHC Class I molecules at all. All cells in the body except red blood cells have these molecules. Therefore, if a Natural Killer Cell comes across a non-red blood cell that is not showing its MHC Class I receptors, it knows the cell has likely been infected and orders it to kill itself. Macrophages then clean up the remains of the cell and the virus inside of it. This process doesn’t just happen while a virus is inside you; these cells are constantly patrolling your cells and looking for signs of stress.
- B Cells, Antibodies, and T Cells — About a week after the infection, your heavy artillery arrives. During that time, Dendritic Cells took their samples from the battlefield to the lymph nodes, found Helper T and Killer T Cells with the specific receptors to match the virus’s antigens, and also activated B Cells. All three of these cell types then cloned themselves, and the B Cells began pumping out tons of Antibodies with receptors to match the virus’s specific antigens. This process took some time. Now, finally, all of these units arrive at the scene in mass to fight the virus. The Antibodies and Killer T Cells really do the trick and help eliminate tons of viruses in your system: millions of Antibodies use their crab-like pincers to clump up tons of viruses for destruction, while Killer T Cells look inside the “display windows” and help eliminate tons of infected cells.
- Healing — The Antibodies, Helper T, and Killer T Cells really help swing the tide when fighting a virus. Once the threat has been neutralized, it’s time for the immune system to power down. If it doesn’t, it will seriously hurt you. Just like the sequencing of an immune response is a chain reaction, so too is the winding down process. Fewer Cytokines being released from your dead cells leads to a halt in inflammation and the production of other immune cells. Most existing fighters kill themselves once the job is done, and Macrophages eat them up and clean up other debris. Slowly but surely, you begin to feel better.
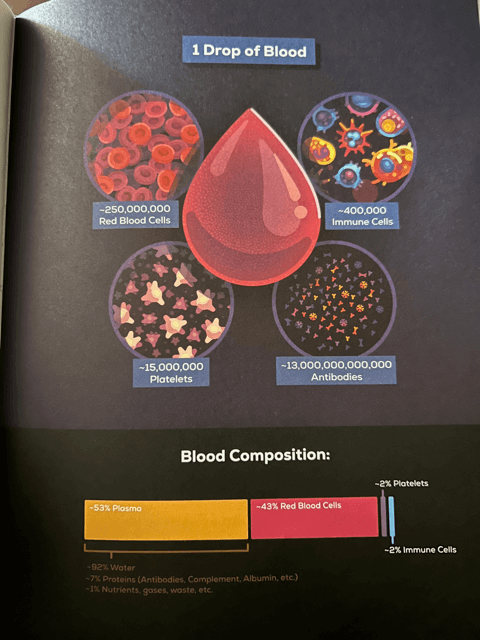
- Immune: Memory Cells — Why is it that we only face a particular sickness or disease one time in our lives? The answer is Memory Cells. After contracting and fighting off a disease, we gain a variety of Memory B Cells and Memory T Cells that “remember” the pathogen and its antigens and can spring to action the moment they sense it in your system. This is one of the most important functions of your immune system. It takes an enormous amount of energy and resources for your body to fight off a pathogen, especially a virus. These Memory Cells act like a shortcut and allow your immune system to skip many of the steps outlined in this book. These Memory Cells patrol your body, create and store Antibodies for the exact pathogen that it already conquered, and attack immediately if the disease ever enters your body again. In fact, every drop of blood contains 13 million, million Antibodies ready to strike any pathogen you’ve already faced. In most cases, you won’t even notice that the same pathogen entered your body; the Memory Cells will remove the threat before it even has a chance to get started.
When it comes to bacteria, we have tons of good medications like antibiotics to help our immune system fight these infections. Why is that not the case with viruses? A lot of it comes down to how bacteria and viruses are structured and how they operate.
In some ways, bacteria are built very differently than our own cells, and antibiotics can take advantage of those differences. Penicillin, for example, works by blocking bacteria’s ability to make cell walls. As a bacterium tries to grow and multiply, it needs to produce more cell walls, and penicillin has a shape that basically interrupts this process and therefore prevents the bacteria from making more of itself. We are able to safely use penicillin because our cells don’t have cell walls — our cells are lined with membranes. Therefore, penicillin harms bacteria without hurting our own cells. Other antibiotics take advantage of similar differences between bacteria and our own cells to eliminate the threat.
Viruses are a different story. We have medications that can help kill viruses — the problem is that those drugs will harm our own cells in the process. Because they spend so much time hiding inside of our cells, it’s hard to use medication to kill viruses without causing a lot of damage to our cells. Even if we try to use drugs to attack the virus outside of our cells, we run into problems. When fighting viruses, drugs are basically a last resort — something to be tried only when the patient is in dire condition.
Enter Vaccines. Vaccines are one of the most effective ways to prevent a certain virus. How do they work? Vaccines are basically weakened versions of a virus or pathogen made in a lab that we can inject into our bodies to protect us against an attack from the real, more dangerous thing. They work because they safely provoke all of the steps of an immune response documented in this book, ultimately concluding with the creation of Memory Cells. When you hear “Vaccine,” think Memory Cells. The two go hand-in-hand, as the goal of a Vaccine is to create Memory Cells inside of you that will remember the pathogen for the rest of your life. Vaccines cause just enough havoc to trigger the right immune response without levying all of the damage that a full-blown attack does.
For example, when Mom tells you to get the latest flu shot before winter, it’s because scientists have studied flu strains circulating on the other side of the world and predicted which one is most likely to hit us. The flu vaccine is then developed in a lab to help your body build immunity to that predicted strain. Given how easily viruses can spread — often floating through the air and entering your body with a single breath through your lungs — getting vaccinated is smart. You make yourself immune to something that has a high probability of entering your vicinity.
Overall, vaccines are good preventative tools that should be used, especially to help kids develop Memory Cells that will make them immune to some of the most dangerous diseases in the world.
🔟 Immune System Hiccups
Although your immune system handles the vast majority of threats without a hitch, there are times where it falters or is simply overmatched. Things like HIV and AIDS, allergies, autoimmune diseases, and cancer are examples of when your immune system is just a little bit off.
In the case of HIV and AIDS, the virus is always one step ahead of the immune system. We are incredibly lucky that this horrible virus is not easy to contract. It doesn’t float through the air or live on surfaces — instead, it is transferred via bodily fluids like blood or intense contact through sexual intercourse. Most HIV infections happen through sexual contact. The primary way this virus causes problems is by infiltrating the Lymph Nodes and knocking out your Helper T Cells, which are critical to your immune response because they play a major role in activating Killer T Cells and B Cells.
The virus is incredibly good at jumping from cell to cell and remaining hidden inside of cells, and it also mutates very quickly. By the time your Adaptive Immune System prepares Killer T Cells and Antibodies to attack the antigens of the HIV virus, it has already changed its genetics and become a slightly different version of itself. Still, your immune system finds a way to mount an attack — but by the time it is ready to strike, most of the virus has changed.
This cycle goes on and on for years and years. As the years go on, you slowly lose more and more Helper T Cells until, one day, your Adaptive Immune System just collapses. At this point, the virus multiplies explosively because there’s nothing to stop it. This leads to AIDS, which basically means that your Adaptive Immune System is out of order. Hundreds of pathogens and cancers that your body normally wiped out without a problem every day now become dangerous and lethal. As a result, the leading causes of death for people with AIDS are various forms of cancer and bacterial or viral infections. Thankfully, modern medicine has turned HIV into a manageable disease.
Allergies, on the other hand, are a case where your immune system is too aggressive. As mentioned throughout this book, the immune system is incredibly powerful and has the potential to hurt you badly, or kill you, if it misfires or is out of sync. Allergies are an example of that. Being allergic means that the immune system massively overreacts to something that isn’t dangerous. It’s like finding a bug in your living room and calling the military to wipe out your city with nuclear weapons. Allergies are no joke — you can die of anaphylactic shock, often within minutes if not treated with epinephrine.
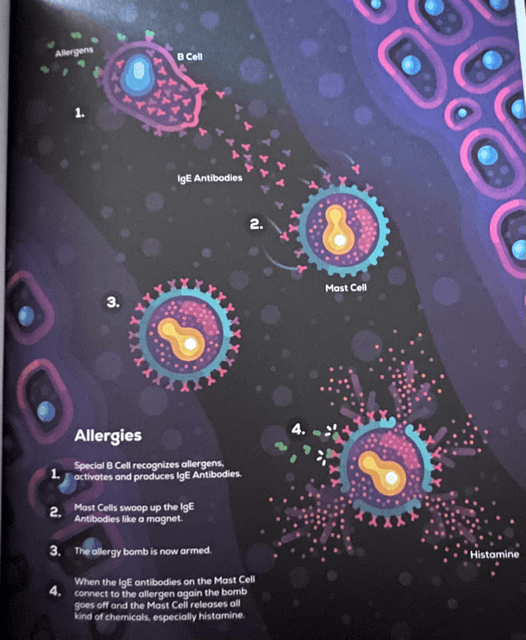
In the case of Allergies, your immune system mistakenly identifies a harmless substance — like pollen, peanuts, or pet dander — as a threat. These substances are called Allergens, which are just antigens — short pieces of protein. What’s wild is that you can suddenly become allergic to things that you’ve eaten all your life. In short, when you eat something like peanuts for the first time, special B Cells in your body recognize the Allergens and produce a variety of Antibodies called IgE Antibodies. Something called a Mast Cell swoops up the IgE Antibodies like a magnet, creating “allergy bombs” just under the surface of your skin and in places like your lungs and gut.
When you are exposed to the Allergen again at a later date, the IgE Antibodies on the Mast Cell connect to the Allergen and the bomb explodes — the Mast Cell releases all of its chemicals, especially Histamine. Histamine causes all of the classic allergy symptoms: inflammation, redness, sneezing, swelling, itching, and more. But what’s worse is that, in severe cases, Histamine can cause the smooth muscles of your lungs to tighten and make breathing hard or even impossible. After the Mast Cells explode, two other types of immune cells jump in to keep the allergy reaction going: Basophils and Eosinophils. Basophils are like backup Mast Cells — they also release Histamine and other chemicals that make the reaction stronger. Eosinophils come in a while later and act like cleanup crews, but instead of calming things down, they can actually make the swelling and damage worse if they get too excited. Eosinophils are a big reason why allergic reactions like runny noses, sneezing, and swelling can stick around longer than you’d expect. Once they arrive, they keep the inflammation going, even after the initial allergy trigger is gone.
What about Autoimmune Diseases? This is another example of your immune system misfiring a bit, this time on your own cells. At its core, the immune system’s job is to identify what is self and what is other. When it finds something that isn’t you, it attacks. Autoimmune Diseases occur when your immune system mistakenly labels your own cells as other and turns on you. These are a variety of these conditions, and the symptoms usually are more annoying and inconvenient than deadly — although the chronic inflammation that results from your immune system constantly attacking is not good at all. An example of an Autoimmune Disease is Type 1 Diabetes, which occurs when your immune cells destroy your pancreas cells and prevent them from making insulin. Somebody with Type 1 Diabetes has to inject lab-made insulin for the rest of their life.
Some people are more likely to get Autoimmune Diseases because of their genes, but genetics aren’t the only factor. Another way it happens is through something called Molecular Mimicry, which occurs when a virus or bacteria enters your body carrying antigens that closely resemble the ones on your own cells. (Yes, your own cells also have antigens — pieces of protein or “tiny ID tags” — on their surface.) To fight the invader, the immune system goes through all of the steps outlined in this book — but because the Helper T Cells, Killer T Cells, B Cells, and Antibodies that are produced in mass are specifically designed to fight an antigen that closely resembles your own cells, they end up also targeting, binding onto, and killing some of your own cells. As with all infections, once the pathogen is eliminated Memory Cells are created to remember that antigen and attack the enemy if it ever returns. The problem in this case is that many — not all — of our own cells have a similar antigen, so the immune system is in a constant state of attack thinking that your cells are the enemy. We continue to replenish our cells as they die, so it’s a never-ending cycle that leads to chronic inflammation and other symptoms like fatigue, pain, and damage to organs.
Finally, we have Cancer. In a nutshell, cancer is when cells in a certain part of your body begin to grow and multiply uncontrollably. There are two major categories of cancer: Tumors and Liquid Cancers. Tumors occur when cancer cells form in solid tissue, like your lungs, muscles, brain, etc. Liquid Cancers affect your blood, bone marrow, lymph, etc. and usually start in the bone marrow. These include leukemia, lymphoma, and multiple myeloma. Leukemia, or blood cancer, is often used as a catchall for these kinds of cancers. With Liquid Cancers, cancer cells travel and multiply within the bloodstream and Lymphatic System — the superhighways your immune cells use to move around. With tumors, the cancer usually starts as a cluster of fast-growing cells in one place — like a rogue village — which can eventually break off and spread to the rest of the body.
One thing to know about cancer right away: Any cell in your body can become cancerous, and getting cancer is not a matter of if, but when. Cancer will kill you if something else doesn’t first. That’s because cancer starts when the genetic code (DNA) inside your cells becomes damaged — usually over many years of normal wear and tear from everyday living. Your body is constantly making new cells to replace old ones that have died. Every time this replication process happens, your genetic code (DNA) is copied over — and sometimes, tiny copying errors called Mutations sneak through. Most mutations are harmless or get fixed. But over time, a few can slip past the body’s repair systems. If certain key genes are affected, those errors can turn a normal cell into a cancer cell. To become cancerous, a cell usually needs three types of mutations:
- Rapid Growth — When you’re an embryo, you’re made up of a small clump of cells. Your Oncogenes monitor the growth and proliferation of a cell, and some of them are very active in the process of turning you from a single cell to an organism with trillions of cells. These rapid growth genes are turned off once you’ve become a fully formed tiny body. But if a mutation years later causes these genes to turn on again, it leads to rapid cell replication. This is the first ingredient of cancer: uncontrolled growth.
- Fixing Errors — Your Tumor Suppressor Genes normally fix DNA errors when cells copy themselves during the replication process. If a mutation disables these genes, damaged cells keep replicating with broken DNA code. This is the second ingredient of cancer: an inability to fix errors.
- Refusal to Die — Finally, your cells normally end their own lives in a process called Apoptosis. This is normal and is what cells do when they’ve performed their duties and have come to the end of their life cycle. It’s also what they do when they sense that something is wrong with them. If a mutation prevents a cell from self-destructing, faulty cells like cancer can live on indefinitely. That’s the third ingredient: immortality.
If a cell collects all three of these mutations, it can become cancerous. And once cancer arrives, it tends to multiply very fast thanks to the Oncogenes being turned on. Think of cancer as a whole new organism living inside of you — an organism that grows and fights your healthy cells for space and nutrients. This is how it kills you: It eventually gets so big and spread out that your organs and cells don’t have any space or resources to survive. It overtakes you.
What’s interesting about cancer is that all of us have cancer cells inside of us right now, but our immune system is very good at taking them out before they become a problem (this process likely occurred while you were reading this). Unfortunately, all it takes is one bad cell to ruin someone’s life. Here’s a look at the three phases of the Cancer vs. Immune System battle:
- First Response — At first, cancer cells grow out of control and build their own messy, dangerous little city — “Tumor Town” — inside your body. They steal nutrients, wreck the neighborhood, and attract attention from the immune system. The body responds by sending in its version of building inspectors and police — immune cells like Killer T Cells and Natural Killer Cells — who identify the rogue construction and start tearing it down. If everything goes right, the tumor is crushed before it can grow further.
- Escalation — Even if most of Tumor Town is destroyed, one sneaky cancer cell might survive — a master of disguise that keeps multiplying in secret. As the immune system keeps attacking, it accidentally trains cancer to get better at hiding. Some cancer cells learn to shut down the immune system’s weapons by flipping “off switches” on the receptors of the very cells sent to destroy them. The result is a dangerous game of hide-and-seek, where the cancer is mutating and evolving to stay one step ahead.
- Spreading — Now, the cancer cells have rebuilt Tumor Town with fake permits and roadblocks that keep police and building inspectors (immune cells) out. They’ve created a hidden world where they can grow undisturbed, spreading quietly and taking over more space. At this point, they’re immune to the immune system and can even spread/travel to other parts of the body (called metastasis), damaging vital organs along the way. Eventually, cancer overwhelms your body’s systems by using up all the space and resources, causing them to shut down. This is what kills you. When a cancer spreads, the situation becomes very, very bad.
In the end, cancer is largely a byproduct of getting older. The longer we live, the more chances our cells have to make mistakes when copying their DNA — and some of those mistakes can lead to cancer. Over time, these mutations can add up, especially if our immune system becomes less effective at catching and destroying damaged cells. While not everyone will develop cancer in their lifetime, the risk increases significantly as we age.


At its core, the immune system is a tool to distinguish self from non-self. Viruses, harmful bacteria, and parasites — all of which can be considered “pathogens” — have been a threat to multicellular life for more than 500 million years, when the first multicellular organisms first appeared. Multicellular beings — like humans and animals — make ideal hosts for these microscopic organisms, who want to invade and take advantage of the resources and nutrients available to them. In order to survive, multicellular beings had to develop sophisticated immune systems to detect and eliminate these threats, which are happening constantly.
The immune system we rely on today is the product of millions of years of evolutionary refinement. Its primary function is to recognize what belongs in the body and what doesn’t. When it identifies something in the body that’s not you — like a virus or harmful bacteria — it attacks it using an army of soldiers and weapons. How does it determine self vs. non-self? If bacteria have been around for billions of years, why haven’t they evolved mechanisms to completely baffle your immune system’s detection capabilities? The answer to these questions lies in the way that bacteria, viruses, and other pathogens are built. Although these microorganisms can build all kinds of different proteins to make themselves look and behave differently to avoid detection, they still need the very basic proteins in order to function. Think of a car — you can mix parts and colors to produce different cars, but you still need wheels and an engine. Your immune cells have receptors that can identify these very basic parts (called Antigens) that are common in most harmful pathogens. A bacteria’s flagella is a good example: the flagella is a tail-like structure made up of proteins that help the bacteria move around. Other than sperm, human cells don’t have a flagella. Therefore, when your immune cells recognize the proteins that make a flagella, they know to kill the organism. This is just one example of how the immune system identifies what doesn’t belong.
Ultimately, the immune system’s goal is to maintain homeostasis — internal balance — within the body. The symptoms you experience during a cold, such as fever or inflammation, are not caused by the virus or bad bacteria itself but by your immune system’s attacks on these bad actors that have invaded your body.
An interesting note about the immune system and pregnancy: when sperm enter a woman’s body, they are recognized as “other” and are attacked by immune system cells like macrophages and neutrophils. Of the 200 million sperm that enter, only a few hundred make it into the fallopian tubes and have a chance to fertilize the egg. Most are eliminated along the way due to the woman’s acidic vaginal environment, immune system, or simply getting off course. This helps explain why getting pregnant is hard.
A similar situation unfolds in the case of organ transplants. The process of installing an organ from one person’s body into another is often very challenging because of the immune system. The immune system labels the new organ, and the cells that make up the organ, as “other” and attacks relentlessly. This is why, unfortunately, many people who receive an organ donation have to spend the rest of their lives taking medication that suppresses the immune system, which leaves them vulnerable to other infections (i.e. bacteria, viruses, cancer).

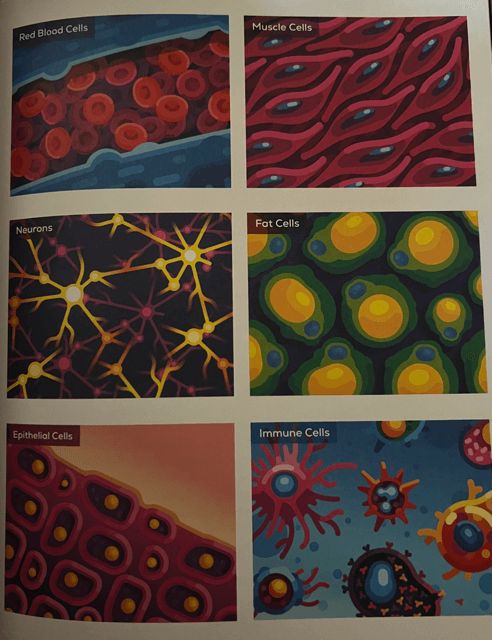
To understand the immune system, you first have to understand cells. We have about 40 trillion cells and 40 trillion helpful bacteria in, and on, our body. About 36 trillion of those bacteria live in our gut and help us digest food; the other four trillion are on our skin, in our lungs and mouth, and in our eye fluid. Cells come in all kinds of types and are jam packed with millions of proteins, which are created when amino acids are linked together in specific sequences. The instructions for assembling these proteins are stored in the DNA found in each cell’s nucleus. Your DNA is a super long strand that consists of smaller sections, your genes, and each gene is the instruction for one protein. The process of building a protein happens in two main steps:
- The DNA is transcribed into a messenger molecule called mRNA
- The mRNA then exits the nucleus and travels to a part of the cell called the ribosome, where it’s translated into a chain of amino acids that folds into a functional protein.
Proteins come in all shapes and sizes, and cells can use them to both build almost anything and communicate with other cells. Think of cells as “protein robots” that are guided by sequences of interactions between proteins. These “protein robots” (i.e. cells) are blind, deaf, and stupid, so how do they know where to go and what to do? Well, cells are covered with receptors that proteins and other things outside of the cell can bind to in order to cause the cell to behave in certain ways. Half of a receptor is inside the cell, half of it is outside the cell. When a certain protein binds to the outer half of the receptor, it sends a signal inside and causes a certain behavior from the cell. For example, when a cytokine — a protein released by immune cells to call for backup at an infection site — binds to the receptor of another immune cell, it steers that cell to the scene. This is the way cells communicate and are able to do things that make sense.
Our immune system is made up of two different realms — the Innate Immune System and the Adaptive Immune System.
The Innate Immune System contains all of the defenses you were born with and can be deployed immediately after an invasion occurs. Its weapons are not tailored to any specific bacteria, virus, or any other pathogen — instead, they try to be effective across a wide range of common invaders. For example, it doesn’t have specific weapons against specific types of bacteria like E.coli; but it does have weapons against bacteria in general. The Innate Immune System is your first line of defense and does most of the actual fighting — in fact, most of the billions of soldiers in your immune system are part of the Inmate Immune System. In addition to fighting bad actors, it has to make crucial decisions after an invasion has occurred: How dangerous is this threat? What kind of enemy is attacking? Are more heavy weapons needed? If your Innate Immune System thinks the threat is big enough, it has the power to activate a second line of defense to join the fight: the soldiers of the Adaptive Immune System.
The Adaptive Immune System contains highly specialized super cells that coordinate and support your first line of defenders in the Innate Immune System. Think of these cells as super soldiers that can come to the rescue if the Innate Immune System is having trouble with a particular bacteria or virus and calls for help. Your Adaptive Immune System knows every possible intruder in the universe and has an answer for every single possible microorganism that exists. It possesses the largest library in the universe, and the library is filled with detailed profiles of every current and future possible enemy. Unlike the Innate Immune System, your Adaptive Immune System is not born with these capabilities; they need to be trained and refined over many years, hence the name “adaptive.” In fact, a weak Adaptive Immune System is part of the reason babies and old people are more likely to die from diseases than people in their middle years.
These two realms are interconnected and play off of each other in a beautifully complex way. The Adaptive Immune System is very powerful in its own right, but its main function is to support the Innate Immune System, which is your first line of defense. If a particular bacterium or virus causes too much trouble for your Innate Immune System at the initial site of the infection, it activates The Adaptive Immune System via Dendritic Cells.
Most harmful bacteria we encounter aren’t much of a problem. Our immune system recognizes their basic protein parts (Antigens) easily and deals with them without much of an issue every single day. Your real enemies are a small group of elite bacteria and viruses that have found ways to overcome your defenses. These are known as pathogens, a term for any microorganism that can make you sick or cause disease. If something makes you sick — whether it’s a virus, bacterium, fungus, or parasite — it’s considered a pathogen.
Let’s start with bacteria. Bacteria are among the oldest living things on this planet. Emphasis on living; these things are alive, unlike viruses. Like your cells, these are single-celled protein robots that come in a wide variety of shapes and sizes based on their DNA. They have evolved over billions of years and have been very successful on Earth. Bacteria are everywhere! We have 36 trillion in our gut (mostly helpful) and another four trillion on our skin. In one gram of plaque on your teeth, there are more than 7 billion bacteria.
Bacteria view us as a great place to sustain life and reproduce. Because they are single-cell organisms, they can reproduce extremely quickly, which is what makes them difficult for your immune system to deal with. They can also change their genetics at any time, which can also cause problems for our immune system. We are absolutely covered in bacteria — but, fortunately, most bacteria are harmless to us. In fact, some even work with us to keep harmful bacteria away, and trillions of them live in our gut, where they primarily help us break down and digest food. These are “friendly” bacteria. And make no mistake, we wouldn’t be alive without some of these friendly bacteria.
If successful, harmful bacteria that invade us can cause a wide variety of diseases and death. They are especially good at entering your body through cuts, where our protective barrier (the skin) has been breached. Antibiotics have certainly helped us fight bacterial diseases, which used to be one of the leading killers of humans. In 1941, 82% of people who incurred bacterial infections of the blood died. Unfathomably, this means a scratch and a tiny bit of dirt might mean the end of your life. Today, in developed countries, less than 1% of these kinds of infections cause death.
Now let’s discuss Viruses. Unlike bacteria, viruses are dead — there is nothing alive about them. In a nutshell, they are made of a couple lines of genetic code and a few proteins. This is why they must infect living things to stick around. Infecting living things is their No. 1 goal, and if all living things on the planet died, they would die as well because there would be nothing to infect. They can’t reproduce or do anything without first infecting a living thing, like a human cell. Unbelievably, there are actually billions and billions and billions of viruses in the world, but only about 200 of them have the proteins necessary to attach to human cells and infect us. Thank goodness.
How does a virus infect us? First it gets inside of us. Rather than breaking through the skin, the most common way it gets inside of us is through our lungs as we breathe in air that a virus is hanging out in (think about someone with the flu coughing near you). Once inside, it usually binds to one of the Epithelial Cells that line the protective mucous layer inside our lungs and takes the cell over by transferring its genetic material. This transfer of genetic material turns the cell into a virus-producing machine. At some point, the cell is completely filled up with viruses. The cell then dies and bursts open, releasing hundreds of new viruses that go out and repeat this same process with cells nearby. Before you know it, the virus has become a raging wildfire inside of your body. And this is a defining feature of these things: Nothing multiplies, and mutates (changes), as fast as a virus does.
Influenza (the flu) and COVID-19 are examples of viruses. The Spanish Flu killed 40 million people in 1918-19. Because viruses like these mutate and multiply so quickly — and because their tactics are different than bacteria — the immune response to a bacterial infection and a viral infection are much different.
Whether it’s harmful bacteria, a virus, a parasite, or even cancer, these pathogens all have one thing in common: they want to destroy your cells. If they manage to get inside, multiply, and spread, they can hijack or kill enough cells to overtake your body, which manifests as serious illness or even death. The immune system’s mission is to protect your cells — and therefore, protect you — by detecting, attacking, and eliminating anything that poses a threat. In this way, every illness or disease comes down to what’s happening with your cells.
How does your immune system recognize and begin attacking these pathogens? Every pathogen — whether it’s bacteria or a virus — has something called Antigens, which are basically small pieces of protein on the surface of the pathogen that serve as “tiny ID tags.” Antigens are really, really important for two main reasons: (i.) they are what your immune system uses to identify exactly what the pathogen is, and (ii.) your Adaptive Immune System uses the Antigens to activate and make the correct Antibodies, Helper T Cells, and Killer T Cells that can lock onto and destroy that specific pathogen. Even our own cells have these “tiny ID tags,” and when our immune system weapons accidentally lock onto our Self-Antigens, it usually leads to Autoimmune Diseases.
For a harmful bacteria or pathogen to become a problem for you, it first has to find a way to break through your skin. Other than your gut, which is made for and ruled by about 36-40 trillion helpful bacteria that your body has welcomed in, the skin is the second most populated place on your body in terms of bacteria. You have about 10 billion bacteria on your skin at all times, some good and some bad. Fortunately for us, our skin is an outstanding protective barrier. Think of it as a dead, salty desert that’s hostile to most microbes, or The Desert Kingdom of the Skin.
The skin is basically made up of hundreds of layers of skin cells that are constantly being regenerated by our skin stem cells. The skin stem cells lie underneath, or deeper in the epidermis, and crank out new skin cells, which form the very bottom layers of your skin and slowly move up over time. As they are pushed up, these living skin cells get coated in a fatty layer of “passive antibiotics” that protect you from harmful enemies. By the time the skin cells reach the surface layers, they’re dead. All of the skin you see in the mirror is dead skin, made up of up to 50 layers of dead skin cells. As this dead layer of skin is damaged and used during your daily life, it is constantly being shed and replaced by new skin cells that are moving up from below. It takes skin between 30-50 days to completely turn over, and you shed about 40,000 dead skin cells every second! That’s important because when your dead skin flakes away, harmful bacteria crawling on it go with it. When you wipe away some dust, a lot of what you’re wiping up is your own dead skin cells.
The skin also becomes salty when you sweat, which harmful bacteria don’t like. As mentioned earlier, we have 10 billion bacteria crawling on our skin at all times — most of these bacteria are not harmful and actually do us a favor by taking up real estate from harmful bacteria. Finally, another great benefit of the skin is that it’s basically immune to viruses, which can only infect living cells — and the surface of your skin consists only of dead cells. Nothing to infect here!
Overall, the skin is an outstanding barrier. In fact, the skin is so effective as a defense perimeter that pathogens usually don’t have much of a shot at getting inside us here, as long as we don’t cut ourselves and open up an opportunity. Instead, most infections that we deal with throughout our lives enter the body elsewhere, like the nose, mouth, eyes, lungs, digestive tract, and reproductive tract. These are the most vulnerable areas on your body because they are where your insides directly interact with the outside world (i.e. breathing in stuff, eating food, etc.). All kinds of stuff that isn’t YOU comes into your body from these areas. As protection, they are lined with what is called Mucosa — or a mucous layer. Think of this as a giant swampland that provides a layer of protection for our insides. It’s the “skin” of our insides. We can call it The Swamp Kingdom of the Mucosa.
The immune system has to behave differently in these mucous layers than it does when you have a breach of the skin. Why? It can’t afford to launch a full-blown immune response to every little thing that comes into your body from breathing or eating, for example. It has to walk a fine line between being too aggressive and too relaxed. If it attacked every little thing that entered, you would have constant inflammation. Not good.
We think of mucous as only being in the nose, but all of these vulnerable areas — the mouth, nose, eyes, respiratory tract (lungs), digestive tract (gut), and reproductive tract — are lined with a layer of it. Just like the skin, mucous contains built-in defense systems that make it difficult for harmful bacteria to get deeper inside of us. For one, the stuff is slimy and is very difficult for bacteria to swim through. Second, it’s loaded with salt, enzymes, and special substances that starve bacteria to death by sponging up critical nutrients that they need. Third, it is saturated with special Antibodies that are ready to “mark” harmful bacteria for destruction by other immune cells if needed.
Depending on the location, mucous behaves a little differently — the mucous layer in your lungs has a very different job than the mucous layer in your gut, and so on. Because it’s fascinating, let’s look at the gut (intestines) as an example. When you eat something, your saliva starts to break it down. After the food is swallowed, it heads to the ocean of acid in your stomach, which breaks the food down and is also where many bacteria on the food die due to being submerged in literal acid. After the stomach, the journey continues to the intestines, where 90% of the nutrients you need to survive are absorbed. Here in the gut is where we find about 36 trillion bacteria that are essential to our survival — they help us digest the food further and produce vitamins that we can’t produce ourselves.
Because of all that bacteria (some good, some bad) that sit directly above the mucous layer of your gut, the immune system of the gut acts semi-independently of the immune system guarding the rest of your body. The mucous layer of the gut is always being breached by some of those bacteria, and your immune system has to constantly determine friend or foe. It does this by having immune cells like Macrophages, B Cells, Dendritic Cells, and Antibodies that behave a little differently than they do in other areas of the body. I won’t get into the details. But together, these cells and Antibodies kill a lot of bacteria in the gut that try to cross the mucous layer — in fact, around 30% of your poop consists of dead (or still alive) bacteria. Also, the last thing you want is a big immune response that leads to inflammation in the gut. Inflammation means more fluid in the gut, which is what causes diarrhea.
Another interesting area to look at is the lungs, where a mucous layer lines the respiratory tract and protects you from all kinds of things. The lungs are where you are most vulnerable to infection from a virus, although bacteria can get in here as well. That’s because you inhale a couple thousand gallons of air through your lungs every day, and there are all kinds of virus, bacteria, fungi, and other stuff hanging out in the air around you. Think about when someone with a cold coughs near you — the stuff in that cloud of air moves quickly and some of it can get into your lungs. Overall, some of the most dangerous pathogenic viruses use the lungs as their main entry point.
Say you cut your knee sliding during an adult softball game. What happens when your skin is opened up and harmful bacteria on you and in the surrounding environment are able to walk right in? To help understand what happens during a bacterial infection, it’s helpful to look at the body as a huge continent with trillions of civilians (cells). A war breaks out, and the cells of the Innate Immune System are the first soldiers sent to fight the enemy. Below is a very simple look at how your Innate Immune System works while fighting a bacterial infection. The immune system’s process for fighting a virus is different and will be covered in a separate takeaway.
- Cytokines — The damage caused by whatever caused your skin to open up kills many of your civilians right away. When your immune cells come across other dead cells or harmful bacteria, they raise the alarm by releasing Cytokines, which are small proteins that convey information to other cells. The cytokines carry the message: “Danger! Enemies are around! Come help!” Immune cells like macrophages and neutrophils “smell” the cytokines and follow their trace to the battlefield. The more cytokines that are released by your immune cells, the bigger the threat. In rare circumstances, your immune cells will release tons of cytokines when the battle is over or when there’s no threat at all. Because cytokines signal the need for an immune response, your cells go crazy attacking things for no reason, eventually causing inflammation and tissue damage. This process is common in autoimmune diseases, where your immune system mistakes you for the enemy.
- Macrophages — Your Innate Immune System responds to the Cytokines immediately by sending Macrophages, the largest immune system cells in the body, to the scene. These cells are huge, much bigger than the average cell — if the normal cell is a human, a Macrophage is a rhino. Macrophages, known as Great Eaters, eat dead cells and living bad bacteria, clear debris from the battlefield, and start the repair process. They swallow as many enemies as they can, but they can’t eliminate the threat by themselves. They release more cytokines and call for backup.
- Neutrophils — Neutrophil cells travel from the blood to the scene to assist. The most abundant immune cell in your blood, they immediately begin hunting and eating bacteria, but they’re much less careful about it because they die fairly quickly. They are the “crazy suicidal Spartan warriors” of the immune system. They eat, throw acid, and kill themselves to create deadly traps. Their goal is to go as hard as they can, and they’re not too concerned about damaging helpful allies. Many of them explode when they’re done, and the chemicals they release are harmful to bad bacteria. Our own healthy civilian cells are sort of afraid of Neutrophils because they are so aggressive. Every day, even without an infection, 100 billion of these warrior cells voluntarily die and are immediately replaced with new ones. The puss that oozes from your cut is the dead bodies of millions of Neutrophils that fought to the death for you, mixed in with dead civilian cells and bacteria.
- Platelets — While all of this is going on, the wound on your skin has developed a bit of a crust. This is your platelets closing it off. Platelets are blood cells that lump together and form a sticky substance that clogs the wound. Their goal is to stop blood loss and prevent more intruders from entering. This enables fresh skin cells to slowly start closing the hole.
- Inflammation — Macrophages and Neutrophils fighting at the scene, as well as cells that have been killed in battle, send signals that cause inflammation, which is a crucial defense process and causes the area around your cut or injury to swell up. Inflammation occurs when your blood vessels open up and let warm fluid stream into the battlefield. Imagine this as floodgates opening up, where blood carrying all sorts of helpful immune cell reinforcements comes rushing in very quickly to defend the injured or infected area. When this happens, the extra fluid causes you to experience some swelling in the area. Inflammation is the universal response of your immune system to any sort of breach or damage. No matter if you burn yourself, cut yourself, or get a bruise. No matter if bacteria or viruses infect your nose, lungs, or gut. Damage or danger — perceived or real — causes inflammation. Inflammation is the red swelling and itching from an insect bite, the sore throat when you have a cold. Interestingly, chronic inflammation is terrible for you; in fact, it’s an underlying cause of about 50% of deaths today. Inflammation is also a big reason for many COVID and pneumonia deaths; when the virus infects people’s lungs, it causes a rush of fluid to the area, and the swelling makes it hard to breathe and get enough oxygen. But in the context of a typical immune response, inflammation is very helpful in three main ways:
- It restricts an infection to an area and stops it from spreading
- It helps remove damaged and dead tissue
- It serves as an expressway to get your immune cells and attack proteins directly to the site of infection
- Complement Proteins — This rush of fluid to the scene caused by inflammation carries a silent killer into the battle zone: Complement Proteins from The Complement System. These proteins are super tiny and float passively in your blood until an infection occurs and one of them is activated. When one of them is activated at the scene of an infection, it causes a chain reaction that leads to the activation of others around it. Soon, an army of millions of these proteins are activated and become a weapon for your immune system. What do they do? Three things: (i.) They “mark” bacteria by coating their surface, making it easier for your Macrophages and Neutrophils to locate, grab, and eat them; (ii.) they call for backup by sending chemical signals to recruit more immune cells to the area; (iii.) they literally “rip holes” in the bacteria, causing them to “bleed out.” Complement Proteins play a big role in creating a very fast Innate Immune System response. Where there’s inflammation, there’s Complement Proteins.
In most cases, this is where the battle ends. All bacteria are killed. Your cut and infection heal up. All is well. But we’ll assume a certain species of harmful bacteria is somehow finding a way to reproduce and evade your Innate Immune System soldiers. Your Macrophages and Neutrophils keep fighting, but the inflammation continues to rise and huge numbers of civilian cells are dying. What happens now? This is when the Adaptive Immune System gets involved. Below is a very high-level look at how this system responds (more detailed information about how some of this unfolds is in the next takeaway):
- Dendritic Cells — When a certain species of harmful bacteria is able to evade your immune system and reproduce quickly in your body, Dendritic Cells have to make a decision about whether or not to call for help and activate the very powerful Adaptive Immune System. Dendritic Cells belong to the Innate Immune System, and they are crucial. As the initial battle at the scene of an infection is taking place, they lurk in the background and assess the situation. They go around and take “samples” of dead enemies. This process involves crunching a bacterium into a pile of Antigens, which are basically the proteins it’s made up of.
- MHC Class II Molecules — Once the bacterium is reduced to a pile of antigens, Dendritic Cells load the antigens onto a molecule that lives inside them called MHC Class II. These molecules look like an empty hot dog bun, and the antigen is loaded onto them like a wiener. Once loaded with wieners (antigens), the MHC Class II molecules move to the surface of the Dendritic Cell so they can “present” the bacteria’s antigens to Helper T Cells of the Adaptive Immune System.
- The Lymphatic System — But first, the Dendritic Cells have to get to the Helper T Cells using the Lymphatic System. For cells, the human body is an enormous continent. The Lymphatic System provides a way for your immune cells to traverse this huge continent. This system is primarily responsible for draining excess fluid called Lymph from your muscles and tissues and delivering it back to the blood so it can recirculate. This process prevents you from swelling up like a balloon over time as the battle is waging. It transports lymph using a network of superhighways (vessels) that your immune cells can also use to travel through the body, and it’s the system the Dendritic Cells use in this case to transport their antigen samples to the Helper T Cells.
- Lymph Nodes — Located throughout your body are about 600 “megacities” called Lymph Nodes. Lymph must pass through these megacities before it can rejoin blood circulation. The lymph nodes are also where immune cells from the Innate Immune System, like Dendritic Cells, meet up with immune cells from the Adaptive Immune System, like Helper T Cells. In this scenario, the Dendritic Cells take their MHC Class II antigen hot dogs to the nearest lymph node stations to find Helper T Cells that have receptors that match the bacterium’s antigens. The exact receptor for the particular antigen that the Dendritic Cells are presenting is not always found right away. Instead, Helper T Cells will literally rearrange their genes to manufacture a receptor that matches the exact antigen of the harmful bacteria.
- T Cells and Antibodies — Once Helper T Cells with the right receptor for the specific antigen wieners it is carrying, more of them are created. Additional weapons built specially to hunt and kill the particular bacterium — like Antibodies and Killer T Cells — are also created. Once created in the lymph nodes, Helper T Cells race over to the battlefield and use special cytokine signals to motivate extremely tired Macrophages. It’s as if they give these soldiers a second wind; a jolt of energy that inspires them to get back to ferociously killing bacteria. Not long after, millions of Antibodies and Killer T Cells arrive on the scene. Antibodies are super soldiers created by B Cells that act as assassins specifically designed to eliminate the exact species of bacteria or virus in your system. They do this primarily by “marking” the enemy for destruction by other immune cells. What was once a brutal battle for days now turns into a one-sided slaughter, as your Adaptive Immune System comes to the rescue.
- Healing — If all goes well, this is where the battle ends. The last bacterium is eaten, Neutrophils and Helper T Cells begin to kill themselves now that victory has been won, Macrophages clean up the debris by eating dead cells before also killing themselves, excess fluid is carried away through the Lymphatic System, inflammation subsides, and young civilian cells take the place of the fallen. Healing is happening. From a human perspective, you notice that the swelling is gone and the wound, injury, or cold feels better.
- Immune: Memory Cells — A critical final piece to this sequence is Memory Cells, which are created after the war has been won. When you hear that you’re immune to a disease, it means you have living Memory T Cells and Memory B Cells roaming around your body that “remember” a specific enemy’s antigens and can easily eliminate the threat if it ever returns later in your life. This is why you normally will not suffer the same sickness or disease twice.
1️⃣ The Immune System: Self vs. Non Self
At its core, the immune system is a tool to distinguish self from non-self. Viruses, harmful bacteria, and parasites — all of which can be considered “pathogens” — have been a threat to multicellular life for more than 500 million years, when the first multicellular organisms first appeared. Multicellular beings — like humans and animals — make ideal hosts for these microscopic organisms, who want to invade and take advantage of the resources and nutrients available to them. In order to survive, multicellular beings had to develop sophisticated immune systems to detect and eliminate these threats, which are happening constantly.
The immune system we rely on today is the product of millions of years of evolutionary refinement. Its primary function is to recognize what belongs in the body and what doesn’t. When it identifies something in the body that’s not you — like a virus or harmful bacteria — it attacks it using an army of soldiers and weapons. How does it determine self vs. non-self? If bacteria have been around for billions of years, why haven’t they evolved mechanisms to completely baffle your immune system’s detection capabilities? The answer to these questions lies in the way that bacteria, viruses, and other pathogens are built. Although these microorganisms can build all kinds of different proteins to make themselves look and behave differently to avoid detection, they still need the very basic proteins in order to function. Think of a car — you can mix parts and colors to produce different cars, but you still need wheels and an engine. Your immune cells have receptors that can identify these very basic parts (called Antigens) that are common in most harmful pathogens. A bacteria’s flagella is a good example: the flagella is a tail-like structure made up of proteins that help the bacteria move around. Other than sperm, human cells don’t have a flagella. Therefore, when your immune cells recognize the proteins that make a flagella, they know to kill the organism. This is just one example of how the immune system identifies what doesn’t belong.
Ultimately, the immune system’s goal is to maintain homeostasis — internal balance — within the body. The symptoms you experience during a cold, such as fever or inflammation, are not caused by the virus or bad bacteria itself but by your immune system’s attacks on these bad actors that have invaded your body.
An interesting note about the immune system and pregnancy: when sperm enter a woman’s body, they are recognized as “other” and are attacked by immune system cells like macrophages and neutrophils. Of the 200 million sperm that enter, only a few hundred make it into the fallopian tubes and have a chance to fertilize the egg. Most are eliminated along the way due to the woman’s acidic vaginal environment, immune system, or simply getting off course. This helps explain why getting pregnant is hard.
A similar situation unfolds in the case of organ transplants. The process of installing an organ from one person’s body into another is often very challenging because of the immune system. The immune system labels the new organ, and the cells that make up the organ, as “other” and attacks relentlessly. This is why, unfortunately, many people who receive an organ donation have to spend the rest of their lives taking medication that suppresses the immune system, which leaves them vulnerable to other infections (i.e. bacteria, viruses, cancer).
2️⃣ Cells: Protein Robots
To understand the immune system, you first have to understand cells. We have about 40 trillion cells and 40 trillion helpful bacteria in, and on, our body. About 36 trillion of those bacteria live in our gut and help us digest food; the other four trillion are on our skin, in our lungs and mouth, and in our eye fluid. Cells come in all kinds of types and are jam packed with millions of proteins, which are created when amino acids are linked together in specific sequences. The instructions for assembling these proteins are stored in the DNA found in each cell’s nucleus. Your DNA is a super long strand that consists of smaller sections, your genes, and each gene is the instruction for one protein. The process of building a protein happens in two main steps:
- The DNA is transcribed into a messenger molecule called mRNA
- The mRNA then exits the nucleus and travels to a part of the cell called the ribosome, where it’s translated into a chain of amino acids that folds into a functional protein.
Proteins come in all shapes and sizes, and cells can use them to both build almost anything and communicate with other cells. Think of cells as “protein robots” that are guided by sequences of interactions between proteins. These “protein robots” (i.e. cells) are blind, deaf, and stupid, so how do they know where to go and what to do? Well, cells are covered with receptors that proteins and other things outside of the cell can bind to in order to cause the cell to behave in certain ways. Half of a receptor is inside the cell, half of it is outside the cell. When a certain protein binds to the outer half of the receptor, it sends a signal inside and causes a certain behavior from the cell. For example, when a cytokine — a protein released by immune cells to call for backup at an infection site — binds to the receptor of another immune cell, it steers that cell to the scene. This is the way cells communicate and are able to do things that make sense.
3️⃣ Two Parts of Your Immune System
Our immune system is made up of two different realms — the Innate Immune System and the Adaptive Immune System.
The Innate Immune System contains all of the defenses you were born with and can be deployed immediately after an invasion occurs. Its weapons are not tailored to any specific bacteria, virus, or any other pathogen — instead, they try to be effective across a wide range of common invaders. For example, it doesn’t have specific weapons against specific types of bacteria like E.coli; but it does have weapons against bacteria in general. The Innate Immune System is your first line of defense and does most of the actual fighting — in fact, most of the billions of soldiers in your immune system are part of the Inmate Immune System. In addition to fighting bad actors, it has to make crucial decisions after an invasion has occurred: How dangerous is this threat? What kind of enemy is attacking? Are more heavy weapons needed? If your Innate Immune System thinks the threat is big enough, it has the power to activate a second line of defense to join the fight: the soldiers of the Adaptive Immune System.
The Adaptive Immune System contains highly specialized super cells that coordinate and support your first line of defenders in the Innate Immune System. Think of these cells as super soldiers that can come to the rescue if the Innate Immune System is having trouble with a particular bacteria or virus and calls for help. Your Adaptive Immune System knows every possible intruder in the universe and has an answer for every single possible microorganism that exists. It possesses the largest library in the universe, and the library is filled with detailed profiles of every current and future possible enemy. Unlike the Innate Immune System, your Adaptive Immune System is not born with these capabilities; they need to be trained and refined over many years, hence the name “adaptive.” In fact, a weak Adaptive Immune System is part of the reason babies and old people are more likely to die from diseases than people in their middle years.
These two realms are interconnected and play off of each other in a beautifully complex way. The Adaptive Immune System is very powerful in its own right, but its main function is to support the Innate Immune System, which is your first line of defense. If a particular bacterium or virus causes too much trouble for your Innate Immune System at the initial site of the infection, it activates The Adaptive Immune System via Dendritic Cells.
4️⃣ Bacteria, Viruses, and Antigens
Most harmful bacteria we encounter aren’t much of a problem. Our immune system recognizes their basic protein parts (Antigens) easily and deals with them without much of an issue every single day. Your real enemies are a small group of elite bacteria and viruses that have found ways to overcome your defenses. These are known as pathogens, a term for any microorganism that can make you sick or cause disease. If something makes you sick — whether it’s a virus, bacterium, fungus, or parasite — it’s considered a pathogen.
Let’s start with bacteria. Bacteria are among the oldest living things on this planet. Emphasis on living; these things are alive, unlike viruses. Like your cells, these are single-celled protein robots that come in a wide variety of shapes and sizes based on their DNA. They have evolved over billions of years and have been very successful on Earth. Bacteria are everywhere! We have 36 trillion in our gut (mostly helpful) and another four trillion on our skin. In one gram of plaque on your teeth, there are more than 7 billion bacteria.
Bacteria view us as a great place to sustain life and reproduce. Because they are single-cell organisms, they can reproduce extremely quickly, which is what makes them difficult for your immune system to deal with. They can also change their genetics at any time, which can also cause problems for our immune system. We are absolutely covered in bacteria — but, fortunately, most bacteria are harmless to us. In fact, some even work with us to keep harmful bacteria away, and trillions of them live in our gut, where they primarily help us break down and digest food. These are “friendly” bacteria. And make no mistake, we wouldn’t be alive without some of these friendly bacteria.
If successful, harmful bacteria that invade us can cause a wide variety of diseases and death. They are especially good at entering your body through cuts, where our protective barrier (the skin) has been breached. Antibiotics have certainly helped us fight bacterial diseases, which used to be one of the leading killers of humans. In 1941, 82% of people who incurred bacterial infections of the blood died. Unfathomably, this means a scratch and a tiny bit of dirt might mean the end of your life. Today, in developed countries, less than 1% of these kinds of infections cause death.
Now let’s discuss Viruses. Unlike bacteria, viruses are dead — there is nothing alive about them. In a nutshell, they are made of a couple lines of genetic code and a few proteins. This is why they must infect living things to stick around. Infecting living things is their No. 1 goal, and if all living things on the planet died, they would die as well because there would be nothing to infect. They can’t reproduce or do anything without first infecting a living thing, like a human cell. Unbelievably, there are actually billions and billions and billions of viruses in the world, but only about 200 of them have the proteins necessary to attach to human cells and infect us. Thank goodness.
How does a virus infect us? First it gets inside of us. Rather than breaking through the skin, the most common way it gets inside of us is through our lungs as we breathe in air that a virus is hanging out in (think about someone with the flu coughing near you). Once inside, it usually binds to one of the Epithelial Cells that line the protective mucous layer inside our lungs and takes the cell over by transferring its genetic material. This transfer of genetic material turns the cell into a virus-producing machine. At some point, the cell is completely filled up with viruses. The cell then dies and bursts open, releasing hundreds of new viruses that go out and repeat this same process with cells nearby. Before you know it, the virus has become a raging wildfire inside of your body. And this is a defining feature of these things: Nothing multiplies, and mutates (changes), as fast as a virus does.
Influenza (the flu) and COVID-19 are examples of viruses. The Spanish Flu killed 40 million people in 1918-19. Because viruses like these mutate and multiply so quickly — and because their tactics are different than bacteria — the immune response to a bacterial infection and a viral infection are much different.
Whether it’s harmful bacteria, a virus, a parasite, or even cancer, these pathogens all have one thing in common: they want to destroy your cells. If they manage to get inside, multiply, and spread, they can hijack or kill enough cells to overtake your body, which manifests as serious illness or even death. The immune system’s mission is to protect your cells — and therefore, protect you — by detecting, attacking, and eliminating anything that poses a threat. In this way, every illness or disease comes down to what’s happening with your cells.
How does your immune system recognize and begin attacking these pathogens? Every pathogen — whether it’s bacteria or a virus — has something called Antigens, which are basically small pieces of protein on the surface of the pathogen that serve as “tiny ID tags.” Antigens are really, really important for two main reasons: (i.) they are what your immune system uses to identify exactly what the pathogen is, and (ii.) your Adaptive Immune System uses the Antigens to activate and make the correct Antibodies, Helper T Cells, and Killer T Cells that can lock onto and destroy that specific pathogen. Even our own cells have these “tiny ID tags,” and when our immune system weapons accidentally lock onto our Self-Antigens, it usually leads to Autoimmune Diseases.
5️⃣ The Desert Kingdom of the Skin & The Swamp Kingdom of the Mucosa
For a harmful bacteria or pathogen to become a problem for you, it first has to find a way to break through your skin. Other than your gut, which is made for and ruled by about 36-40 trillion helpful bacteria that your body has welcomed in, the skin is the second most populated place on your body in terms of bacteria. You have about 10 billion bacteria on your skin at all times, some good and some bad. Fortunately for us, our skin is an outstanding protective barrier. Think of it as a dead, salty desert that’s hostile to most microbes, or The Desert Kingdom of the Skin.
The skin is basically made up of hundreds of layers of skin cells that are constantly being regenerated by our skin stem cells. The skin stem cells lie underneath, or deeper in the epidermis, and crank out new skin cells, which form the very bottom layers of your skin and slowly move up over time. As they are pushed up, these living skin cells get coated in a fatty layer of “passive antibiotics” that protect you from harmful enemies. By the time the skin cells reach the surface layers, they’re dead. All of the skin you see in the mirror is dead skin, made up of up to 50 layers of dead skin cells. As this dead layer of skin is damaged and used during your daily life, it is constantly being shed and replaced by new skin cells that are moving up from below. It takes skin between 30-50 days to completely turn over, and you shed about 40,000 dead skin cells every second! That’s important because when your dead skin flakes away, harmful bacteria crawling on it go with it. When you wipe away some dust, a lot of what you’re wiping up is your own dead skin cells.
The skin also becomes salty when you sweat, which harmful bacteria don’t like. As mentioned earlier, we have 10 billion bacteria crawling on our skin at all times — most of these bacteria are not harmful and actually do us a favor by taking up real estate from harmful bacteria. Finally, another great benefit of the skin is that it’s basically immune to viruses, which can only infect living cells — and the surface of your skin consists only of dead cells. Nothing to infect here!
Overall, the skin is an outstanding barrier. In fact, the skin is so effective as a defense perimeter that pathogens usually don’t have much of a shot at getting inside us here, as long as we don’t cut ourselves and open up an opportunity. Instead, most infections that we deal with throughout our lives enter the body elsewhere, like the nose, mouth, eyes, lungs, digestive tract, and reproductive tract. These are the most vulnerable areas on your body because they are where your insides directly interact with the outside world (i.e. breathing in stuff, eating food, etc.). All kinds of stuff that isn’t YOU comes into your body from these areas. As protection, they are lined with what is called Mucosa — or a mucous layer. Think of this as a giant swampland that provides a layer of protection for our insides. It’s the “skin” of our insides. We can call it The Swamp Kingdom of the Mucosa.
The immune system has to behave differently in these mucous layers than it does when you have a breach of the skin. Why? It can’t afford to launch a full-blown immune response to every little thing that comes into your body from breathing or eating, for example. It has to walk a fine line between being too aggressive and too relaxed. If it attacked every little thing that entered, you would have constant inflammation. Not good.
We think of mucous as only being in the nose, but all of these vulnerable areas — the mouth, nose, eyes, respiratory tract (lungs), digestive tract (gut), and reproductive tract — are lined with a layer of it. Just like the skin, mucous contains built-in defense systems that make it difficult for harmful bacteria to get deeper inside of us. For one, the stuff is slimy and is very difficult for bacteria to swim through. Second, it’s loaded with salt, enzymes, and special substances that starve bacteria to death by sponging up critical nutrients that they need. Third, it is saturated with special Antibodies that are ready to “mark” harmful bacteria for destruction by other immune cells if needed.
Depending on the location, mucous behaves a little differently — the mucous layer in your lungs has a very different job than the mucous layer in your gut, and so on. Because it’s fascinating, let’s look at the gut (intestines) as an example. When you eat something, your saliva starts to break it down. After the food is swallowed, it heads to the ocean of acid in your stomach, which breaks the food down and is also where many bacteria on the food die due to being submerged in literal acid. After the stomach, the journey continues to the intestines, where 90% of the nutrients you need to survive are absorbed. Here in the gut is where we find about 36 trillion bacteria that are essential to our survival — they help us digest the food further and produce vitamins that we can’t produce ourselves.
Because of all that bacteria (some good, some bad) that sit directly above the mucous layer of your gut, the immune system of the gut acts semi-independently of the immune system guarding the rest of your body. The mucous layer of the gut is always being breached by some of those bacteria, and your immune system has to constantly determine friend or foe. It does this by having immune cells like Macrophages, B Cells, Dendritic Cells, and Antibodies that behave a little differently than they do in other areas of the body. I won’t get into the details. But together, these cells and Antibodies kill a lot of bacteria in the gut that try to cross the mucous layer — in fact, around 30% of your poop consists of dead (or still alive) bacteria. Also, the last thing you want is a big immune response that leads to inflammation in the gut. Inflammation means more fluid in the gut, which is what causes diarrhea.
Another interesting area to look at is the lungs, where a mucous layer lines the respiratory tract and protects you from all kinds of things. The lungs are where you are most vulnerable to infection from a virus, although bacteria can get in here as well. That’s because you inhale a couple thousand gallons of air through your lungs every day, and there are all kinds of virus, bacteria, fungi, and other stuff hanging out in the air around you. Think about when someone with a cold coughs near you — the stuff in that cloud of air moves quickly and some of it can get into your lungs. Overall, some of the most dangerous pathogenic viruses use the lungs as their main entry point.
6️⃣ An Immune System Response: Bacterial Infection
Say you cut your knee sliding during an adult softball game. What happens when your skin is opened up and harmful bacteria on you and in the surrounding environment are able to walk right in? To help understand what happens during a bacterial infection, it’s helpful to look at the body as a huge continent with trillions of civilians (cells). A war breaks out, and the cells of the Innate Immune System are the first soldiers sent to fight the enemy. Below is a very simple look at how your Innate Immune System works while fighting a bacterial infection. The immune system’s process for fighting a virus is different and will be covered in a separate takeaway.
- Cytokines — The damage caused by whatever caused your skin to open up kills many of your civilians right away. When your immune cells come across other dead cells or harmful bacteria, they raise the alarm by releasing Cytokines, which are small proteins that convey information to other cells. The cytokines carry the message: “Danger! Enemies are around! Come help!” Immune cells like macrophages and neutrophils “smell” the cytokines and follow their trace to the battlefield. The more cytokines that are released by your immune cells, the bigger the threat. In rare circumstances, your immune cells will release tons of cytokines when the battle is over or when there’s no threat at all. Because cytokines signal the need for an immune response, your cells go crazy attacking things for no reason, eventually causing inflammation and tissue damage. This process is common in autoimmune diseases, where your immune system mistakes you for the enemy.
- Macrophages — Your Innate Immune System responds to the Cytokines immediately by sending Macrophages, the largest immune system cells in the body, to the scene. These cells are huge, much bigger than the average cell — if the normal cell is a human, a Macrophage is a rhino. Macrophages, known as Great Eaters, eat dead cells and living bad bacteria, clear debris from the battlefield, and start the repair process. They swallow as many enemies as they can, but they can’t eliminate the threat by themselves. They release more cytokines and call for backup.
- Neutrophils — Neutrophil cells travel from the blood to the scene to assist. The most abundant immune cell in your blood, they immediately begin hunting and eating bacteria, but they’re much less careful about it because they die fairly quickly. They are the “crazy suicidal Spartan warriors” of the immune system. They eat, throw acid, and kill themselves to create deadly traps. Their goal is to go as hard as they can, and they’re not too concerned about damaging helpful allies. Many of them explode when they’re done, and the chemicals they release are harmful to bad bacteria. Our own healthy civilian cells are sort of afraid of Neutrophils because they are so aggressive. Every day, even without an infection, 100 billion of these warrior cells voluntarily die and are immediately replaced with new ones. The puss that oozes from your cut is the dead bodies of millions of Neutrophils that fought to the death for you, mixed in with dead civilian cells and bacteria.
- Platelets — While all of this is going on, the wound on your skin has developed a bit of a crust. This is your platelets closing it off. Platelets are blood cells that lump together and form a sticky substance that clogs the wound. Their goal is to stop blood loss and prevent more intruders from entering. This enables fresh skin cells to slowly start closing the hole.
- Inflammation — Macrophages and Neutrophils fighting at the scene, as well as cells that have been killed in battle, send signals that cause inflammation, which is a crucial defense process and causes the area around your cut or injury to swell up. Inflammation occurs when your blood vessels open up and let warm fluid stream into the battlefield. Imagine this as floodgates opening up, where blood carrying all sorts of helpful immune cell reinforcements comes rushing in very quickly to defend the injured or infected area. When this happens, the extra fluid causes you to experience some swelling in the area. Inflammation is the universal response of your immune system to any sort of breach or damage. No matter if you burn yourself, cut yourself, or get a bruise. No matter if bacteria or viruses infect your nose, lungs, or gut. Damage or danger — perceived or real — causes inflammation. Inflammation is the red swelling and itching from an insect bite, the sore throat when you have a cold. Interestingly, chronic inflammation is terrible for you; in fact, it’s an underlying cause of about 50% of deaths today. Inflammation is also a big reason for many COVID and pneumonia deaths; when the virus infects people’s lungs, it causes a rush of fluid to the area, and the swelling makes it hard to breathe and get enough oxygen. But in the context of a typical immune response, inflammation is very helpful in three main ways:
- It restricts an infection to an area and stops it from spreading
- It helps remove damaged and dead tissue
- It serves as an expressway to get your immune cells and attack proteins directly to the site of infection
- Complement Proteins — This rush of fluid to the scene caused by inflammation carries a silent killer into the battle zone: Complement Proteins from The Complement System. These proteins are super tiny and float passively in your blood until an infection occurs and one of them is activated. When one of them is activated at the scene of an infection, it causes a chain reaction that leads to the activation of others around it. Soon, an army of millions of these proteins are activated and become a weapon for your immune system. What do they do? Three things: (i.) They “mark” bacteria by coating their surface, making it easier for your Macrophages and Neutrophils to locate, grab, and eat them; (ii.) they call for backup by sending chemical signals to recruit more immune cells to the area; (iii.) they literally “rip holes” in the bacteria, causing them to “bleed out.” Complement Proteins play a big role in creating a very fast Innate Immune System response. Where there’s inflammation, there’s Complement Proteins.
In most cases, this is where the battle ends. All bacteria are killed. Your cut and infection heal up. All is well. But we’ll assume a certain species of harmful bacteria is somehow finding a way to reproduce and evade your Innate Immune System soldiers. Your Macrophages and Neutrophils keep fighting, but the inflammation continues to rise and huge numbers of civilian cells are dying. What happens now? This is when the Adaptive Immune System gets involved. Below is a very high-level look at how this system responds (more detailed information about how some of this unfolds is in the next takeaway):
- Dendritic Cells — When a certain species of harmful bacteria is able to evade your immune system and reproduce quickly in your body, Dendritic Cells have to make a decision about whether or not to call for help and activate the very powerful Adaptive Immune System. Dendritic Cells belong to the Innate Immune System, and they are crucial. As the initial battle at the scene of an infection is taking place, they lurk in the background and assess the situation. They go around and take “samples” of dead enemies. This process involves crunching a bacterium into a pile of Antigens, which are basically the proteins it’s made up of.
- MHC Class II Molecules — Once the bacterium is reduced to a pile of antigens, Dendritic Cells load the antigens onto a molecule that lives inside them called MHC Class II. These molecules look like an empty hot dog bun, and the antigen is loaded onto them like a wiener. Once loaded with wieners (antigens), the MHC Class II molecules move to the surface of the Dendritic Cell so they can “present” the bacteria’s antigens to Helper T Cells of the Adaptive Immune System.
- The Lymphatic System — But first, the Dendritic Cells have to get to the Helper T Cells using the Lymphatic System. For cells, the human body is an enormous continent. The Lymphatic System provides a way for your immune cells to traverse this huge continent. This system is primarily responsible for draining excess fluid called Lymph from your muscles and tissues and delivering it back to the blood so it can recirculate. This process prevents you from swelling up like a balloon over time as the battle is waging. It transports lymph using a network of superhighways (vessels) that your immune cells can also use to travel through the body, and it’s the system the Dendritic Cells use in this case to transport their antigen samples to the Helper T Cells.
- Lymph Nodes — Located throughout your body are about 600 “megacities” called Lymph Nodes. Lymph must pass through these megacities before it can rejoin blood circulation. The lymph nodes are also where immune cells from the Innate Immune System, like Dendritic Cells, meet up with immune cells from the Adaptive Immune System, like Helper T Cells. In this scenario, the Dendritic Cells take their MHC Class II antigen hot dogs to the nearest lymph node stations to find Helper T Cells that have receptors that match the bacterium’s antigens. The exact receptor for the particular antigen that the Dendritic Cells are presenting is not always found right away. Instead, Helper T Cells will literally rearrange their genes to manufacture a receptor that matches the exact antigen of the harmful bacteria.
- T Cells and Antibodies — Once Helper T Cells with the right receptor for the specific antigen wieners it is carrying, more of them are created. Additional weapons built specially to hunt and kill the particular bacterium — like Antibodies and Killer T Cells — are also created. Once created in the lymph nodes, Helper T Cells race over to the battlefield and use special cytokine signals to motivate extremely tired Macrophages. It’s as if they give these soldiers a second wind; a jolt of energy that inspires them to get back to ferociously killing bacteria. Not long after, millions of Antibodies and Killer T Cells arrive on the scene. Antibodies are super soldiers created by B Cells that act as assassins specifically designed to eliminate the exact species of bacteria or virus in your system. They do this primarily by “marking” the enemy for destruction by other immune cells. What was once a brutal battle for days now turns into a one-sided slaughter, as your Adaptive Immune System comes to the rescue.
- Healing — If all goes well, this is where the battle ends. The last bacterium is eaten, Neutrophils and Helper T Cells begin to kill themselves now that victory has been won, Macrophages clean up the debris by eating dead cells before also killing themselves, excess fluid is carried away through the Lymphatic System, inflammation subsides, and young civilian cells take the place of the fallen. Healing is happening. From a human perspective, you notice that the swelling is gone and the wound, injury, or cold feels better.
- Immune: Memory Cells — A critical final piece to this sequence is Memory Cells, which are created after the war has been won. When you hear that you’re immune to a disease, it means you have living Memory T Cells and Memory B Cells roaming around your body that “remember” a specific enemy’s antigens and can easily eliminate the threat if it ever returns later in your life. This is why you normally will not suffer the same sickness or disease twice.
7️⃣ The Adaptive Immune System
The Adaptive Immune System is extremely powerful and complex, and it deserves a closer look as its own takeaway. As mentioned earlier, the Adaptive Immune System contains specialized super soldiers that can help the Innate Immune System deal with a particular species of harmful bacteria or virus that is reproducing quickly and causing problems. The warriors of this system do not come to the battlefield of an infection right away; the Innate Immune System soldiers head to the scene first while Dendritic Cells assess the situation. If the Innate Immune System struggles enough, Dendritic Cells will call on the very powerful Adaptive Immune System for help by taking the pathogen’s antigens to Helper T Cells and B Cells located in your lymph nodes.
Why is our Adaptive Immune System so important? Bacteria have been around for billions of years and possess two features that pose big problems for our immune system: (i.) they are single-cell organisms, which allows them to reproduce extremely fast, and (ii.) they can literally change their own genetics/proteins at any time to evade Innate Immune System soldiers (e.g. Macrophages and Neutrophils). Most bacteria on and around you are harmless or even helpful to you. But these unique abilities of bacteria become a big issue when harmful bacteria looking to take you out get into your body (e.g. E.coli). Your Innate Immune System soldiers (e.g. Macrophages and Neutrophils) usually don’t have any problem recognizing the antigens (i.e. proteins) on these harmful bacteria and killing them; they do it every day without batting an eye. But when a particular species of harmful bacteria possessing antigens that the Innate Immune System is not familiar with gets in and begins reproducing and changing genes, it needs help before things get really bad for you in the form of disease. These bacteria evade detection because they possess antigens (proteins) that the Innate Immune System is not familiar with.
Viruses are arguably even worse. They are able to hide inside of your cells, and they multiply explosively. The Innate Immune System is not very effective at fighting viruses — the best it can really do is buy some time for more effective weapons.
That’s where the Adaptive Immune System comes in. This system has an incredible superpower: it can generate millions of different immune cells, each with receptors specifically designed to recognize specific antigens — which are protein pieces from a virus, bacterium, or other invader. Together, these cells form the largest biological “library” in your body and give you the ability to make weapons for any pathogen the world might throw at you. Even for new threats like COVID-19, your Adaptive Immune System can either find or create (via mixing and matching genes) immune cells with the right receptor — it just takes time. That’s why it’s called “adaptive”: it learns and responds to each new invader, building immunity that can protect you in the future.
Arguably the most important infantry of soldiers in this system are T Cells, which are born in the Bone Marrow but are trained for duty in the Thymus (hence the name “T Cells”). Resting a few inches under your throat, the Thymus is one of the most under-appreciated organs in the human body. One of its big responsibilities is training T Cells; it’s a giant T Cell training academy. T Cells — Both Helper T Cells and Killer T Cells — are born with receptors capable of binding to the antigens (proteins) of bacteria and can mix and match their genetic code to connect to every possible antigen in the universe. This is why you have the ability to recognize and fight any infection known to man.
During an infection, Dendritic Cells from the Innate Immune System can activate the Adaptive Immune System by finding a Helper T Cell that has the exact receptor for the harmful bacteria’s antigen that it is carrying (the MHC Class II “hot dog” from the previous takeaway). When this happens, the Helper T Cell clones itself in your lymph nodes until there are thousands of them ready to fight the bacteria that is causing problems (this is why your lymph nodes swell during a cold). Then they split into two groups. One group heads to the battlefield and takes command. They put tired Macrophages into killer mode using chemical signals. They coordinate things between other fighters. This happens about 5-7 days after the infection. The second group remains in the lymph nodes to help activate your B Cells, which are your living weapons factory.
B Cells originate in the Bone Marrow and are similar to T Cells in that, combined, they have millions of different receptors for millions of antigens that a harmful bacteria might possess. What makes B Cells special and very dangerous is that they produce the most lethal specialized weapon in the immune system: Antibodies. These are receptors on the surface of B Cells that can also be used as weapons against specific antigens that a pathogen possesses. B Cells must undergo rigorous training in the Bone Marrow and are then released into your Lymphatic System in an inactive state. B Cells can only become activated using a two-factor authentication process:
- Step 1 — B Cells hang out in your Lymph Nodes, the “megacities” of the Lymphatic System “superhighway” network. Lymph — which carries away from the battlefield excess fluid from inflammation, antigens (proteins) from dead enemy bacteria, and other “garbage” — passes through the Lymph Nodes. B Cells basically sift through the Lymph looking for antigens that match their receptors. If they find an antigen that matches one of their receptors, they know that there’s an enemy in the body and begin making clones of the exact B Cell with the specific receptor that matches the enemy’s antigen. These cloned B Cells then begin making limited quantities of basic Antibodies while they await the second step.
- Step 2 — Without a second step, these B Cells from step one would die in about a day, figuring the infection must not be that bad. A B Cell becomes FULLY activated when it finds an activated Helper T Cell presenting the same enemy antigen. When this happens, the B Cell knows for sure that something bad is going on and turns into a Plasma Cell that generates highly specialized Antibodies. The Plasma Cell pumps these out at a rate of about 2,000 per second — millions of them are created and sent to the battlefield through the blood and Lymphatic System to fight the pathogen that is causing problems for you. This is one of the most powerful stages of the Adaptive Immune System and can turn the tide of an infection quickly.
So, what are Antibodies? They are your super weapons, the main reason you can survive serious infections. Antibodies are essentially copies of the B Cell’s receptor, designed to stick to one specific antigen on an enemy and flag it for destruction. Think of antibodies as proteins that act like guided missiles. They can neutralize viruses by preventing them from infecting your living cells and “marking” bacteria for cleanup by other immune cells like Macrophages and Neutrophils. Antibodies are tiny in structure and look like little crabs with two pincers and a “cute little butt.” The pincers “pinch” the pathogen’s antigens with a death-grip, and their “cute little butt” makes it easy for other immune cells like Macrophages and Neutrophils to latch on and kill the pathogen. Some Antibodies can team up and bind to multiple pathogens, creating big clumps of enemy bodies ready for execution. This makes it nice and easy for your other immune cells to score “double kills” and “triple kills.”
Killer T Cells are also activated via a two-step verification process and are critical when fighting a virus. Viruses like to hide and replicate inside of your cells. They then cause the cell to burst open, and the viruses go out and repeat the process with other nearby cells. In short, Killer T Cells are able to inspect each of your cells and look inside of them. If they see virus proteins inside the cell, they order the cell to kill itself gently. The cell then folds up, trapping the virus inside, and Macrophages come by to eat the virus.
Once all harmful bacteria are destroyed and the infection is over, most Helper T Cells kill themselves, while a small group of them become Memory T Cells. When you hear that you’re immune to a disease, it means you have living Memory T Cells and Memory B Cells roaming around your body that “remember” a specific enemy and can easily kill it if it ever returns later in your life. This is why you normally will not suffer the same sickness or disease twice.
8️⃣ Underappreciated Organs: The Thymus and Spleen
One of the real dangers of the immune system is that the soldiers that fight your enemies might turn on you. This is a particular concern for T Cells and B Cells. Fortunately, there are “academies” that these cells must pass before they are cleared for duty.
For your body, the worry with both of these powerful cells is that they might be born with a receptor that binds to, and kills, your own friendly cells. This is the cause of most Autoimmune Diseases — our Adaptive Immune System thinks our own cells are enemies and attacks us. For T Cells, the Thymus helps prevent that. It’s an academy that newly born T Cells must pass before being put to use. The biggest exam at the academy involves testing a T Cell’s ability to recognize and connect to the proteins (Self-Antigens) on the surface of our own cells. If it can, it is killed. All in all, 98 of 100 T Cells are killed; just 2% pass the Thymus academy and are cleared for duty. This amounts to about 20 million T Cells cleared to enter your bloodstream and Lymphatic System to fight infections. The dead cadets are eaten by your Macrophages. The Thymus is always shrinking, and by about age 85, it’s almost entirely inactive. Once it’s gone, your body struggles to produce new T Cells, which is a huge hit to your immune system. This is one of the reasons older adults have a hard time battling infections, disease, and cancer.
B Cells must also pass rigorous training before they are cleared for duty and allowed to patrol the body. Their training happens in the Bone Marrow, which is the same place they are born. The training curriculum is very similar to what T Cells go through — the most important test is that they cannot bind to the proteins and molecules on the surface of your own friendly cells. If they prove during training that they can, they are killed.
The Spleen is another under-appreciated organ. It’s basically a large Lymph Node about the size of a peach that does all of the things that your 600+ Lymph Nodes do — but instead of filtering Lymph, its focus is on filtering blood. The Spleen is where your old blood cells are filtered and recycled when their life comes to an end. Your Spleen also stores an emergency reserve of blood, about a cup, which is critical if something bad happens and your body needs extra blood. And that’s not all — it also serves as one of the centers of your immune system soldier cells, a sort of barracks. The Spleen should get more love than it does!
9️⃣ An Immune System Response: Viral Infections
Our immune system can’t rely on the same weapons to fight a viral infection as it uses to fight a bacterial infection. A virus is smaller and usually a lot more difficult to detect than bacteria. They also hide inside of living cells and try to manipulate infected cells to trick the immune system into standing down. Think back to the ancient Greeks and their invasion of Troy. Bacteria are like the warriors who stormed the gates — loud, aggressive, and ready for battle out in the open. Our immune system can usually recognize the threat and handle it fairly easily. Viruses, on the other hand, are like the soldiers hidden inside the Trojan horse — silent, sneaky, and waiting to strike from within once they’re let inside. Viruses can also mutate (change) and multiply much, much more quickly than bacteria.
Pathogenic viruses are truly scary enemies. They hide inside civilian cells and can multiply explosively. At the height of a virus, you can have billions of viruses inside your body. As a result, your immune system has to approach viruses differently than bacteria. It’s complicated, but here’s a simplified and general look at what happens:
- Entry — A virus, like the flu, enters the body usually through the lungs and the air we breathe in. All kinds of viruses and bacteria hang out in the air around us and can enter us this way. The lungs are one of our most vulnerable and exposed areas. As mentioned previously, viruses find it almost impossible to break through the skin.
- Hostile Cell Takeover — Once inside, most viruses die in the mucous layer lining our lungs. But occasionally one (all it takes is one) is able to bind to one of the Epithelial Cells that lines the mucous layer and performs a “hostile takeover” of the cell by transferring its genetic material. This transfer of genetic material turns the cell into a virus-producing machine. Eventually, the cell is completely filled up with viruses. The cell then dies and bursts open, releasing hundreds of new viruses that go out and repeat this same process with cells nearby. If an immune response doesn’t happen quickly, the virus can become a raging wildfire inside of your body.
- Cytokines & Interferons — At this point, the Adaptive Immune System is desperately needed — but it takes time for that system to get going. The Innate Immune System has to buy some time and prevent the virus from spreading like a wildfire. Epithelial Cells that realize they are being invaded by a virus do this by releasing Cytokines, which act as signals for backup. They specifically release a type of emergency Cytokine called Interferons, which warn other cells to shut down protein production. This slows down the spread of the virus considerably by making surrounding cells more aware and resistant to infection.
- Plasmacytoid Dendritic Cells — Viruses love to stay hidden from the immune system, but there comes a point where that’s not possible anymore. When cells die an unnatural death, they release all kinds of Cytokines that warn the rest of the immune system that danger is present. So as more and more cells die from the virus, more Cytokines are released and more inflammation is triggered. Plasmacytoid Dendritic Cells aid this warning process. These are a special type of Dendritic Cell that are very good at detecting viruses and pump out huge amounts of Interferons. This helps warn the rest of the immune system and tells nearby cells to shutdown protein production, which slows the virus down.
- Macrophages and Neutrophils — At this point, a full-blown war is on. Macrophages and Neutrophils of the Innate Immune System rush to the scene and begin doing what they can. But the Innate Immune System is not very good at fighting viruses; it can only really slow the virus down, not stop it. The Adaptive Immune System is desperately needed.
- Pyrogens and Fever — As the war wages on, your immune cells begin to release another kind of Cytokine called Pyrogens that act as a more serious call for help. Pyrogens are chemicals that cause Fever. They tell the brain to crank up the temperature. Why is a Fever important while fighting any kind of pathogen? It creates a body-wide heating response that makes life much harder for pathogens while also enabling your immune cells to fight harder. Think about running outside — it’s much harder to run in warm or hot temperatures versus nice, crisp, cool temperatures. The same thing takes place for pathogens in your body when a Fever occurs. Note — a Fever of 104 degrees Fahrenheit is very dangerous and you should seek immediate medical attention. Your brain cranks up the heat and induces a Fever in two main ways:
- Shivering — Your brain induces shivering, which is just your muscles contracting really quickly. These quick contractions generate heat as a byproduct.
- Closing Blood Vessels — The blood vessels close to the surface of your body contract which reduces the heat that can escape through your skin. This is why you usually feel so cold while being sick or having a Fever. Your brain and body are trying to heat up your core to make things unpleasant on the pathogen.
- Additional Physical Symptoms — The war being fought inside you manifests as the typical symptoms of a flu: Fever, muscle aches, runny nose, sore throat, cough, feeling cold, etc. When you blow your nose, the more colorful your snot, the more Neutrophils have given their life. Another symptom is fatigue and lack of appetite. These symptoms are your body trying to tell you to conserve energy so it can fight the pathogen. An immune response takes a ton of energy. So does digesting food and doing anything but laying in bed. To conserve energy for the fight, you should try to obey these signals by resting and not eating too much.
- MHC Class I Molecules — Back to the battle. Viruses spend most of their time hiding and multiplying inside of your living cells. This is why they are so dangerous and hard to stop. To stop the virus, your immune system has to be able recognize, then kill, your own cells that have been infected. How does this happen? The answer is Major Histocompatibility Complex Class I Molecules (MHC Class I). These molecules act as “display windows,” showing what kind of proteins and antigens are being made inside of a cell. As discussed earlier, cells are filled with millions of proteins. When a virus infects one of your cells, it begins creating virus proteins and antigens. It’s complicated, but MHC Class I molecules basically allow your immune cells to go up to any cell in your body with a nucleus (so, all but red blood cells), peer into a little “display window,” and see what kind of proteins are inside. If the proteins inside look foreign, the cell is deemed infected and killed on the spot.
- Killer T Cells — Killer T Cells are among the most powerful cells in your immune system, and they depend on the MHC Class I molecule described above. Dendritic Cells of the Innate Immune System are able to activate Helper T Cells and Killer T Cells. They have this capability because they are able to present a pathogen’s antigens in both MHC Class I and MHC Class II molecules. The MHC Class I molecule (display window) helps the Dendritic Cell activate the Killer T Cell with the right receptor for the pathogen’s antigens, while the MHC Class II (the hot dog) helps it locate and activate the Helper T Cell with the right receptor.
- “Serial Killing” — Once activated, the Killer T Cell clones itself then heads to the infected area to evaluate your cells. It goes up to the MHC Class I “display windows” on every one of the cells in the area and peers inside. If what it sees inside is not right, it deems the cell infected and tells the cell to kill itself in a controlled manner. By dying in a controlled manner, the virus is carefully trapped inside the dead cell, and Macrophages come by to eat everything up. This T Cell murder spree happens at a mass scale — usually about 10 days after the battle started — as it quickly takes out your infected cells in a process known as “serial killing.”
- Natural Killer Cells — Viruses aren’t stupid. They know that Killer T Cells use MHC Class I molecules to peer inside infected cells and decide whether or not to kill them. As a result, viruses found a way to prevent this “display window” from appearing outside the infected cells, allowing them to stay hidden from Killer T Cells and other immune cells. Enter Natural Killer Cells. These are siblings of Killer T Cells that arrive at the battleground within 2-3 days of infection. Their job? To hunt cells infected by viruses and cancer. How do they do this? They check to see if a cell has MHC Class I molecules at all. All cells in the body except red blood cells have these molecules. Therefore, if a Natural Killer Cell comes across a non-red blood cell that is not showing its MHC Class I receptors, it knows the cell has likely been infected and orders it to kill itself. Macrophages then clean up the remains of the cell and the virus inside of it. This process doesn’t just happen while a virus is inside you; these cells are constantly patrolling your cells and looking for signs of stress.
- B Cells, Antibodies, and T Cells — About a week after the infection, your heavy artillery arrives. During that time, Dendritic Cells took their samples from the battlefield to the lymph nodes, found Helper T and Killer T Cells with the specific receptors to match the virus’s antigens, and also activated B Cells. All three of these cell types then cloned themselves, and the B Cells began pumping out tons of Antibodies with receptors to match the virus’s specific antigens. This process took some time. Now, finally, all of these units arrive at the scene in mass to fight the virus. The Antibodies and Killer T Cells really do the trick and help eliminate tons of viruses in your system: millions of Antibodies use their crab-like pincers to clump up tons of viruses for destruction, while Killer T Cells look inside the “display windows” and help eliminate tons of infected cells.
- Healing — The Antibodies, Helper T, and Killer T Cells really help swing the tide when fighting a virus. Once the threat has been neutralized, it’s time for the immune system to power down. If it doesn’t, it will seriously hurt you. Just like the sequencing of an immune response is a chain reaction, so too is the winding down process. Fewer Cytokines being released from your dead cells leads to a halt in inflammation and the production of other immune cells. Most existing fighters kill themselves once the job is done, and Macrophages eat them up and clean up other debris. Slowly but surely, you begin to feel better.
- Immune: Memory Cells — Why is it that we only face a particular sickness or disease one time in our lives? The answer is Memory Cells. After contracting and fighting off a disease, we gain a variety of Memory B Cells and Memory T Cells that “remember” the pathogen and its antigens and can spring to action the moment they sense it in your system. This is one of the most important functions of your immune system. It takes an enormous amount of energy and resources for your body to fight off a pathogen, especially a virus. These Memory Cells act like a shortcut and allow your immune system to skip many of the steps outlined in this book. These Memory Cells patrol your body, create and store Antibodies for the exact pathogen that it already conquered, and attack immediately if the disease ever enters your body again. In fact, every drop of blood contains 13 million, million Antibodies ready to strike any pathogen you’ve already faced. In most cases, you won’t even notice that the same pathogen entered your body; the Memory Cells will remove the threat before it even has a chance to get started.
When it comes to bacteria, we have tons of good medications like antibiotics to help our immune system fight these infections. Why is that not the case with viruses? A lot of it comes down to how bacteria and viruses are structured and how they operate.
In some ways, bacteria are built very differently than our own cells, and antibiotics can take advantage of those differences. Penicillin, for example, works by blocking bacteria’s ability to make cell walls. As a bacterium tries to grow and multiply, it needs to produce more cell walls, and penicillin has a shape that basically interrupts this process and therefore prevents the bacteria from making more of itself. We are able to safely use penicillin because our cells don’t have cell walls — our cells are lined with membranes. Therefore, penicillin harms bacteria without hurting our own cells. Other antibiotics take advantage of similar differences between bacteria and our own cells to eliminate the threat.
Viruses are a different story. We have medications that can help kill viruses — the problem is that those drugs will harm our own cells in the process. Because they spend so much time hiding inside of our cells, it’s hard to use medication to kill viruses without causing a lot of damage to our cells. Even if we try to use drugs to attack the virus outside of our cells, we run into problems. When fighting viruses, drugs are basically a last resort — something to be tried only when the patient is in dire condition.
Enter Vaccines. Vaccines are one of the most effective ways to prevent a certain virus. How do they work? Vaccines are basically weakened versions of a virus or pathogen made in a lab that we can inject into our bodies to protect us against an attack from the real, more dangerous thing. They work because they safely provoke all of the steps of an immune response documented in this book, ultimately concluding with the creation of Memory Cells. When you hear “Vaccine,” think Memory Cells. The two go hand-in-hand, as the goal of a Vaccine is to create Memory Cells inside of you that will remember the pathogen for the rest of your life. Vaccines cause just enough havoc to trigger the right immune response without levying all of the damage that a full-blown attack does.
For example, when Mom tells you to get the latest flu shot before winter, it’s because scientists have studied flu strains circulating on the other side of the world and predicted which one is most likely to hit us. The flu vaccine is then developed in a lab to help your body build immunity to that predicted strain. Given how easily viruses can spread — often floating through the air and entering your body with a single breath through your lungs — getting vaccinated is smart. You make yourself immune to something that has a high probability of entering your vicinity.
Overall, vaccines are good preventative tools that should be used, especially to help kids develop Memory Cells that will make them immune to some of the most dangerous diseases in the world.
🔟 Immune System Hiccups
Although your immune system handles the vast majority of threats without a hitch, there are times where it falters or is simply overmatched. Things like HIV and AIDS, allergies, autoimmune diseases, and cancer are examples of when your immune system is just a little bit off.
In the case of HIV and AIDS, the virus is always one step ahead of the immune system. We are incredibly lucky that this horrible virus is not easy to contract. It doesn’t float through the air or live on surfaces — instead, it is transferred via bodily fluids like blood or intense contact through sexual intercourse. Most HIV infections happen through sexual contact. The primary way this virus causes problems is by infiltrating the Lymph Nodes and knocking out your Helper T Cells, which are critical to your immune response because they play a major role in activating Killer T Cells and B Cells.
The virus is incredibly good at jumping from cell to cell and remaining hidden inside of cells, and it also mutates very quickly. By the time your Adaptive Immune System prepares Killer T Cells and Antibodies to attack the antigens of the HIV virus, it has already changed its genetics and become a slightly different version of itself. Still, your immune system finds a way to mount an attack — but by the time it is ready to strike, most of the virus has changed.
This cycle goes on and on for years and years. As the years go on, you slowly lose more and more Helper T Cells until, one day, your Adaptive Immune System just collapses. At this point, the virus multiplies explosively because there’s nothing to stop it. This leads to AIDS, which basically means that your Adaptive Immune System is out of order. Hundreds of pathogens and cancers that your body normally wiped out without a problem every day now become dangerous and lethal. As a result, the leading causes of death for people with AIDS are various forms of cancer and bacterial or viral infections. Thankfully, modern medicine has turned HIV into a manageable disease.
Allergies, on the other hand, are a case where your immune system is too aggressive. As mentioned throughout this book, the immune system is incredibly powerful and has the potential to hurt you badly, or kill you, if it misfires or is out of sync. Allergies are an example of that. Being allergic means that the immune system massively overreacts to something that isn’t dangerous. It’s like finding a bug in your living room and calling the military to wipe out your city with nuclear weapons. Allergies are no joke — you can die of anaphylactic shock, often within minutes if not treated with epinephrine.
In the case of Allergies, your immune system mistakenly identifies a harmless substance — like pollen, peanuts, or pet dander — as a threat. These substances are called Allergens, which are just antigens — short pieces of protein. What’s wild is that you can suddenly become allergic to things that you’ve eaten all your life. In short, when you eat something like peanuts for the first time, special B Cells in your body recognize the Allergens and produce a variety of Antibodies called IgE Antibodies. Something called a Mast Cell swoops up the IgE Antibodies like a magnet, creating “allergy bombs” just under the surface of your skin and in places like your lungs and gut.
When you are exposed to the Allergen again at a later date, the IgE Antibodies on the Mast Cell connect to the Allergen and the bomb explodes — the Mast Cell releases all of its chemicals, especially Histamine. Histamine causes all of the classic allergy symptoms: inflammation, redness, sneezing, swelling, itching, and more. But what’s worse is that, in severe cases, Histamine can cause the smooth muscles of your lungs to tighten and make breathing hard or even impossible. After the Mast Cells explode, two other types of immune cells jump in to keep the allergy reaction going: Basophils and Eosinophils. Basophils are like backup Mast Cells — they also release Histamine and other chemicals that make the reaction stronger. Eosinophils come in a while later and act like cleanup crews, but instead of calming things down, they can actually make the swelling and damage worse if they get too excited. Eosinophils are a big reason why allergic reactions like runny noses, sneezing, and swelling can stick around longer than you’d expect. Once they arrive, they keep the inflammation going, even after the initial allergy trigger is gone.
What about Autoimmune Diseases? This is another example of your immune system misfiring a bit, this time on your own cells. At its core, the immune system’s job is to identify what is self and what is other. When it finds something that isn’t you, it attacks. Autoimmune Diseases occur when your immune system mistakenly labels your own cells as other and turns on you. These are a variety of these conditions, and the symptoms usually are more annoying and inconvenient than deadly — although the chronic inflammation that results from your immune system constantly attacking is not good at all. An example of an Autoimmune Disease is Type 1 Diabetes, which occurs when your immune cells destroy your pancreas cells and prevent them from making insulin. Somebody with Type 1 Diabetes has to inject lab-made insulin for the rest of their life.
Some people are more likely to get Autoimmune Diseases because of their genes, but genetics aren’t the only factor. Another way it happens is through something called Molecular Mimicry, which occurs when a virus or bacteria enters your body carrying antigens that closely resemble the ones on your own cells. (Yes, your own cells also have antigens — pieces of protein or “tiny ID tags” — on their surface.) To fight the invader, the immune system goes through all of the steps outlined in this book — but because the Helper T Cells, Killer T Cells, B Cells, and Antibodies that are produced in mass are specifically designed to fight an antigen that closely resembles your own cells, they end up also targeting, binding onto, and killing some of your own cells. As with all infections, once the pathogen is eliminated Memory Cells are created to remember that antigen and attack the enemy if it ever returns. The problem in this case is that many — not all — of our own cells have a similar antigen, so the immune system is in a constant state of attack thinking that your cells are the enemy. We continue to replenish our cells as they die, so it’s a never-ending cycle that leads to chronic inflammation and other symptoms like fatigue, pain, and damage to organs.
Finally, we have Cancer. In a nutshell, cancer is when cells in a certain part of your body begin to grow and multiply uncontrollably. There are two major categories of cancer: Tumors and Liquid Cancers. Tumors occur when cancer cells form in solid tissue, like your lungs, muscles, brain, etc. Liquid Cancers affect your blood, bone marrow, lymph, etc. and usually start in the bone marrow. These include leukemia, lymphoma, and multiple myeloma. Leukemia, or blood cancer, is often used as a catchall for these kinds of cancers. With Liquid Cancers, cancer cells travel and multiply within the bloodstream and Lymphatic System — the superhighways your immune cells use to move around. With tumors, the cancer usually starts as a cluster of fast-growing cells in one place — like a rogue village — which can eventually break off and spread to the rest of the body.
One thing to know about cancer right away: Any cell in your body can become cancerous, and getting cancer is not a matter of if, but when. Cancer will kill you if something else doesn’t first. That’s because cancer starts when the genetic code (DNA) inside your cells becomes damaged — usually over many years of normal wear and tear from everyday living. Your body is constantly making new cells to replace old ones that have died. Every time this replication process happens, your genetic code (DNA) is copied over — and sometimes, tiny copying errors called Mutations sneak through. Most mutations are harmless or get fixed. But over time, a few can slip past the body’s repair systems. If certain key genes are affected, those errors can turn a normal cell into a cancer cell. To become cancerous, a cell usually needs three types of mutations:
- Rapid Growth — When you’re an embryo, you’re made up of a small clump of cells. Your Oncogenes monitor the growth and proliferation of a cell, and some of them are very active in the process of turning you from a single cell to an organism with trillions of cells. These rapid growth genes are turned off once you’ve become a fully formed tiny body. But if a mutation years later causes these genes to turn on again, it leads to rapid cell replication. This is the first ingredient of cancer: uncontrolled growth.
- Fixing Errors — Your Tumor Suppressor Genes normally fix DNA errors when cells copy themselves during the replication process. If a mutation disables these genes, damaged cells keep replicating with broken DNA code. This is the second ingredient of cancer: an inability to fix errors.
- Refusal to Die — Finally, your cells normally end their own lives in a process called Apoptosis. This is normal and is what cells do when they’ve performed their duties and have come to the end of their life cycle. It’s also what they do when they sense that something is wrong with them. If a mutation prevents a cell from self-destructing, faulty cells like cancer can live on indefinitely. That’s the third ingredient: immortality.
If a cell collects all three of these mutations, it can become cancerous. And once cancer arrives, it tends to multiply very fast thanks to the Oncogenes being turned on. Think of cancer as a whole new organism living inside of you — an organism that grows and fights your healthy cells for space and nutrients. This is how it kills you: It eventually gets so big and spread out that your organs and cells don’t have any space or resources to survive. It overtakes you.
What’s interesting about cancer is that all of us have cancer cells inside of us right now, but our immune system is very good at taking them out before they become a problem (this process likely occurred while you were reading this). Unfortunately, all it takes is one bad cell to ruin someone’s life. Here’s a look at the three phases of the Cancer vs. Immune System battle:
- First Response — At first, cancer cells grow out of control and build their own messy, dangerous little city — “Tumor Town” — inside your body. They steal nutrients, wreck the neighborhood, and attract attention from the immune system. The body responds by sending in its version of building inspectors and police — immune cells like Killer T Cells and Natural Killer Cells — who identify the rogue construction and start tearing it down. If everything goes right, the tumor is crushed before it can grow further.
- Escalation — Even if most of Tumor Town is destroyed, one sneaky cancer cell might survive — a master of disguise that keeps multiplying in secret. As the immune system keeps attacking, it accidentally trains cancer to get better at hiding. Some cancer cells learn to shut down the immune system’s weapons by flipping “off switches” on the receptors of the very cells sent to destroy them. The result is a dangerous game of hide-and-seek, where the cancer is mutating and evolving to stay one step ahead.
- Spreading — Now, the cancer cells have rebuilt Tumor Town with fake permits and roadblocks that keep police and building inspectors (immune cells) out. They’ve created a hidden world where they can grow undisturbed, spreading quietly and taking over more space. At this point, they’re immune to the immune system and can even spread/travel to other parts of the body (called metastasis), damaging vital organs along the way. Eventually, cancer overwhelms your body’s systems by using up all the space and resources, causing them to shut down. This is what kills you. When a cancer spreads, the situation becomes very, very bad.
In the end, cancer is largely a byproduct of getting older. The longer we live, the more chances our cells have to make mistakes when copying their DNA — and some of those mistakes can lead to cancer. Over time, these mutations can add up, especially if our immune system becomes less effective at catching and destroying damaged cells. While not everyone will develop cancer in their lifetime, the risk increases significantly as we age.






